In order to achieve the desired therapeutic effect, sufficient active pharmaceutical ingredient needs to reach the site of action. However, many APIs tend to have very low solubility.
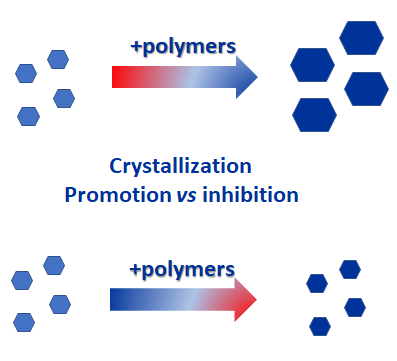
One of the methods to improve their transient solubility and dissolution rate is to use the drug in its amorphous state stabilized in an additives matrix. Many studies have shown that crystallization from supersaturated solutions can be inhibited or slowed down by use of certain additives.
As Taylor et al [1] mentioned back in 2015, the effectiveness of a given polymer is highly dependent on its interaction with the API under investigation. The additive/polymer needs to inhibit crystallization in the solid formulation during storage, as well as in the aqueous environment during dissolution. Taylor et al evaluated the crystallization inhibition effects of several polymers (APAP, HPMC, PAA, HPMCAS, PVPVA, PVP) in the solid state and in aqueous supersaturated solutions by using small scale crystallization reactors. Two of the polymers showed to have the greatest effect on nucleation and were the most effective at inhibiting growth, suggesting possible similarities in their mechanism. The impact of a polymer on the supersaturation profile depends not only on its effect on nucleation, but also on its ability to suppress crystal growth once nuclei have formed. The authors have used the Crystalline instrument with through the vial the particle viewer for growth rate estimations by capturing images every second while precipitation was taking place.

Matzger et al [6] investigated whether solubility of polymers can either speed up or slow down a crystallization process. The authors have tested if insoluble polymers accelerate crystallization and soluble polymers inhibit crystallization. They have found that the same functional groups that promote heteronucleation on an insoluble polymer also promote crystallization inhibition by using a soluble polymer.
Nevertheless, the authors further mention that the ability of water-soluble polymers to inhibit crystallization is more sensitive to the functional group chemistry than the ability of insoluble polymers to accelerate crystallization. Matzger et al used the CrystalBreeder to monitor the crystallization by the changes in solution turbidity. The authors have performed 80 induction times for each crystallization condition in order to assess the effects of the different polymers.
The CrystalBreeder offered Matzger et al an easy, effective and automatic way to perform multiple experiments in the same time. Additionally, the CrystalBreeder with the feedback control functionality enables you to easily detect when the compound is completely dissolved (clear point) and controls crystallization by triggering the next temperature-controlled step (cloud point).
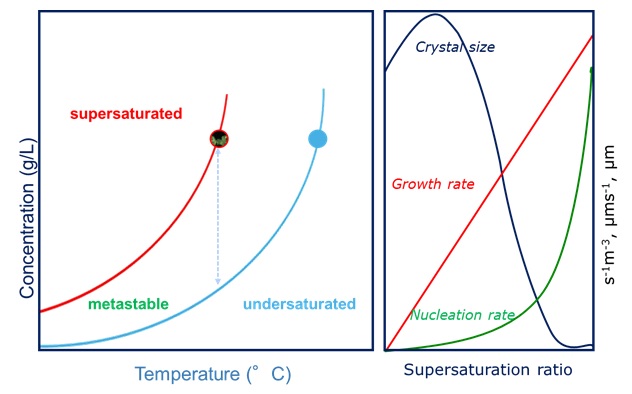
Moreover, one of the strategies to improve filterability in process crystallization is to modify the crystal shape and or size of a crystal form. This can be achieved by selecting a specific solvent or by using additives. Additives preferentially adsorb onto different growing crystal faces and inhibit/promote crystallization in certain directions. Additives may directly influence the size of the metastable zone width (MSZW). Supersaturation is the driving force for both the nucleation and the crystal growth step in a crystallization process. Every solution has a maximum supersaturation limit that is defined as metastable zone width (MSZW). The knowledge of MSZW is very important in terms of designing crystallization processes and obtaining desired crystal sizes, shapes, and purities. For this reason, the supersaturation level should be controlled and the crystallizer has to be operated at an optimum supersaturation level that is approximately half of the width of the metastable zone.
The Crystal16 and Crystalline instruments are the perfect tools for investigating the effect of additives on the crystallization process of an active ingredient.
The Crystal16 combines automation with integrated transmissivity measurement to determine clear (complete dissolution) and cloud (crystallization/precipitation) points resulting in solubility data at an early stage with only a minimal amount of sample. The transmissivity measurement allows one to determine cloud and clear points and thus the MSZW. Using MSZW and the solubility curves, our users are now able to significantly improve their crystallization processes.
The Crystalline instrument with through the vial analytical capabilities like particle visualization allows you to visualize the crystallization process. Crystal shape engineering has never been easier!

References
- N. S. Trasi, K. A. Oucherif, J. D. Litster and L. S. Taylor, CrystEngComm, 2015,17, 1242–1248.
- I. Weissbuch, M. Lahav and L. Leiserowitz, Cryst. Growth Des., 2003, 3, 125–150.
- V. Y. Torbeev, E. Shavit, I. Weissbuch, L. Leiserowitz and M. Lahav, Cryst. Growth Des., 2005, 5, 2190–2196.
- S. Ozaki, I. Kushida, T. Yamashita, T. Hasebe, O. Shirai and K. Kano, J. Pharm. Sci., 2013, 102, 2273–2281.
- S. Kirboga and M. Oner, Colloids Surf., B, 2012, 91, 18 –25.
- D. S. Frank, Q. Zhu, A. J. Matzger, Mol. Pharmaceutics 2019, 16, 8, 3720-3725.
- S. Wu, Kangli Li, J. Gong, BIWIC 2014, 21st International Workshop on Industrial Crystallization
- P. Kongsamai, A. Maneedaeng, C. Flood, J.H ter Horst, A. Flood, Eur. Phys. J. Spec. Top., 2017, 226, 823-835.
Curious to learn more?
Discover our Workflow – CrystalBreeder, Crystal16 and Crystalline instruments – for fully controlled crystallization studies.