
Crystal16
The new industry standard
Fast track your formulation research to success with Crystal16‘s solubility measurements and Crystalline‘s particle visualization cameras
Most crystalline materials are not used directly as powders, but as complex formulations specific for their intended application. This is true for pharmaceutical formulations, as solid doses (pills or capsules) or parenteral formulations (injectable), and for other industries as well, such as agrochemicals, where the final product is usually a sprayable solution.

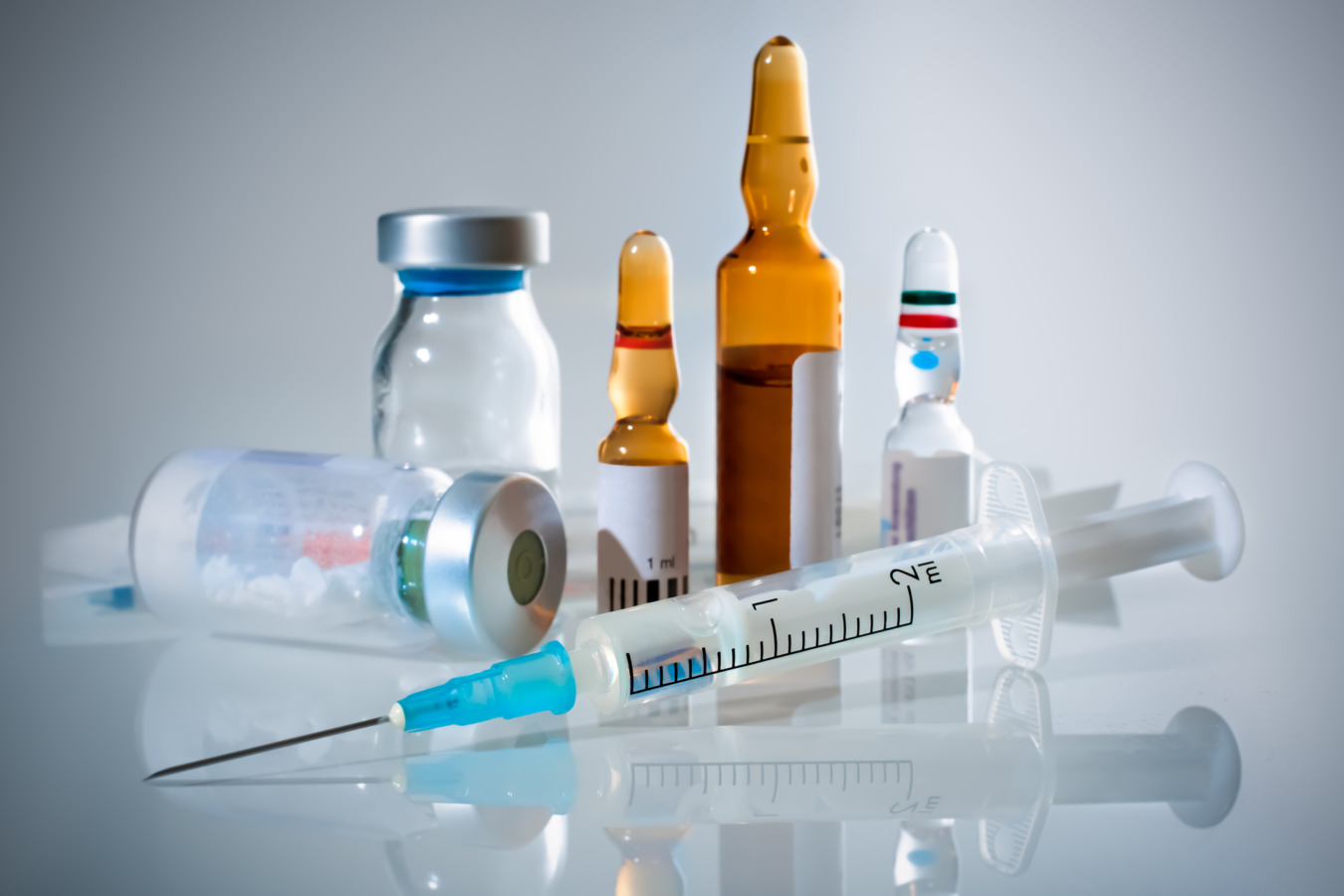
Many drug substances are formulated as parenteral formulations and administrated directly into the systemic circulation in animals and humans. In order to improve their stability and solubility, many parenteral formulations are formulated using a co-solvent. Ampule or vial dosage forms are often co-solvent concentrated formulations. These concentrates are finally diluted before administered to the patient. Nevertheless, there are many cases known where dilution of drug formulations has the potential to generate conditions where drug concentrations are supersaturated.Under these circumstances, drug precipitation/crystallization is likely to take place. Drug precipitation after parenteral administration may cause mechanical or chemical irritation at the injection site, and potentially even more serious systemic effects. The main challenge is that the solubility of an active ingredient in a mixture of solvents may not be linear during dilution. The sooner the temperature dependent solubility of the active ingredient is determined, the better.
Crystal16 can carry solubility measurements fast and easy, and help you avoid vulnerable formulations and mechanical and chemical irritations at injection site.

The Crystalline device can speed up your formulation process significantly, by allowing you to test 8 distinct formulations at 5 mL scale each, and to track their behaviour using the integrated high-resolution cameras. You can also follow your formulations' chemical stability using a Raman probe. This approach gives you early access to crucial information, such as the stability of protein formulations, and allows you to see any changes in your compound in real time.

This application note discusses how to quickly obtain data on the stability of protein formulations, made possible with the use of the real time digital cameras of the Crystalline instrument. It includes a case study on the stability of iysozyme formulations.