Leif-Thore Deck
Doctoral student
Leif-Thore Deck pursues his doctoral studies at ETH Zürich’s Separation Processes Laboratory with Prof. Marco Mazzotti in the field of crystallization applied to pharmaceutical manufacturing. Leif graduated from ETH Zürich in 2020 with an M.Sc. in Chemical and Bioengineering. During his studies, he spent half a year each at the National University of Singapore and at MIT.
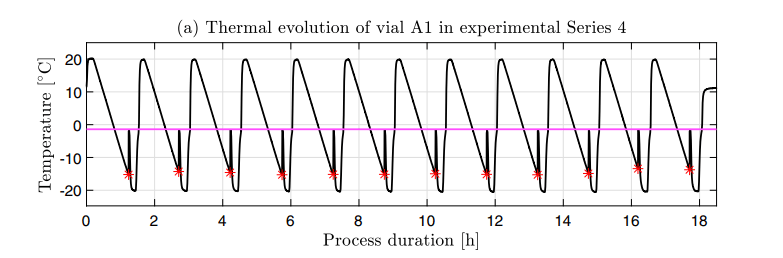
In a research project funded by Janssen J&J, Leif investigates the role of stochastic ice nucleation in pharmaceutical freezing processes via modeling and experiments. Stochastic nucleation poses a major challenge in the design and optimization of pharmaceutical processes that involve freezing in vials. It leads to inherent vial-to-vial variability in thermal evolution that may negatively affect both process behavior and product quality. A majority of biopharmaceuticals, including most commercially available COVID-19 vaccines, undergo freezing or freeze-drying. They thus may benefit from a deepened understanding of ice nucleation.