Solubility-based approach for co-crystallization
Introduction
Within the pharma industry, active ingredients are commonly formulated as solid dosages, however, the properties of the solid form of the actives are not always ideal. Therefore, crystal engineering is often employed to improve properties. Co-crystals are a form of crystal engineering that is gaining an increased level of interest within pharma. The growing body of research has shown to improve physiochemical properties such as solubility, stability, taste masking, dissolution profile, bioavailability, and melting point. Co-crystals are a multi-component complex, similar to salts, solvates, and hydrates, where a second molecule is incorporated into the crystal lattice. However, unlike other complexes, the second molecule must be neutral and not a solvent.
The difference between salt and a co-crystal can be simplified to whether a proton is transferred between the two molecules (salt) or the proton is bonded between the two (co-crystal). To determine whether a salt or co-crystal is being formed a single crystal diffraction should be used.
Solubility approach to screening
Conventional screening methods which use stoichiometric quantities of active and co-former may miss a significant number of potential co-crystals. Using a systematic approach to co-crystal screening utilizing the clear point information generated with the Crystal16, it is possible to improve the probability of finding potential co-crystals as well as construct phase diagrams to better understand the co-crystal landscape.
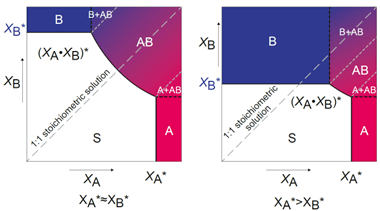
Figure 1 Co-crystal phase diagrams
Carbamazepine is an anticonvulsant used in the treatment of schizophrenia and is a fairly common compound used in research as it has multiple solid forms. Carbamazepine and the artificial sweetener cinnamic acid were used to develop and demonstrate the effectiveness of the systematic approach to cocrystal screening using solubility information generated in the Crystal16. The instrument combines automation with integrated turbidity measurement (transmissivity) to determine clear and cloud points resulting in solubility data at an early stage with only a minimal amount of sample. The Crystal16 can generate solubility curves for four solvents in a short time with less than 100 mg of material (figure 4). By collecting the solubility points of the active ingredient and the coformers, isonicotinamide (INA), nicotinamide (NA), benzamide (BA), 3-nitrobenazamide (NBA), picolinamide (PA) and 2-ethoxybenzamide (EBA)) using the Crystal16’s transmissivity data, van’t Hoff solubility curves can be generated from several concentration Tclear points.
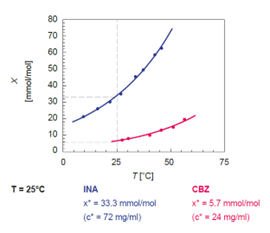
Figure 2
From the solubility curves, one may generate a Tclear for a sample consisting of the saturation concentrations of the individual components. If the solubility temperature of this mixture is greater than the individual components, it indicates the formation of a more stable less soluble form, a co-crystal. Further confirmation of co-crystal formation may be pursued by making use of secondary analysis such as single crystal XRD or a combination of analysis such as XRPD, and DSC/TGMS.
Phase diagrams
Once co-crystals have been created using this systematic method, it is possible to generate a phase diagram with a few extra measurements. Collecting Tclear points for a range of ratios of the two components allows you to identify the ratios where the solubility is dictated by the co-crystal and thus the stable region to operate for scale up and single crystal experiments.
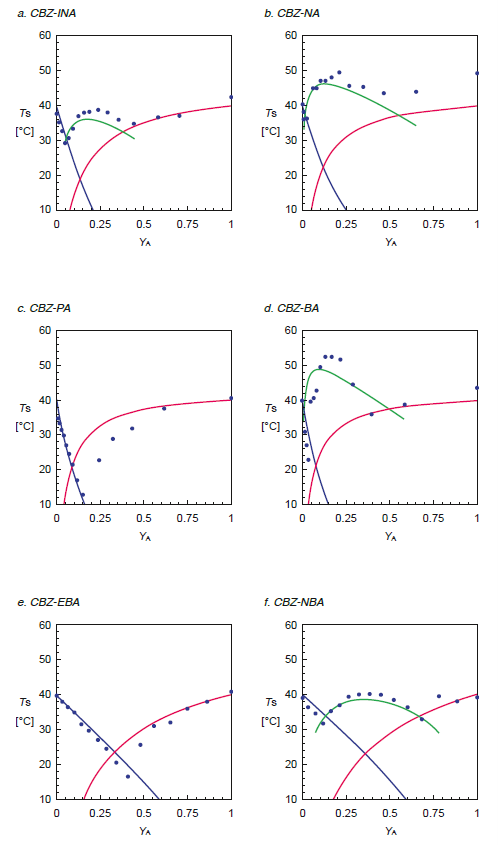
Figure 3 Phase diagrams from co-crystal screen
Conclusion
The Crystal16 instrument enables a systematic approach to co-crystal screening. By making use of the solubility information of the individual components, it is possible to identify a concentration of the two components where the solubility of the co-crystal is the dominant factor, being 10°C greater than the solubility of the components (figure 7). Collection of the solubility data used for this screening method can be done reliably, rapidly, and simply. Using this method and the Crystal16 instrument, cocrystals of carbamazepine with isonicotinamide, nicotinamide, benzamide and 3-nitrobenzamide and of cinnamic acid with isonicotinamide and 3-nitrobenzamide may be easily made. Furthermore, a phase diagram can be constructed by collecting solubility information in a range of ratios of the two components, which is a useful tool in single crystal experiments, as well as scale-up process development.
Acknowledgements
Technobis Crystallization Systems is deeply grateful to Prof. Joop ter Horst for his help with this application note.
References
Thomas Kendall, Sam Stratford, Adam R. Patterson, Ruth A. Lunt, Dyanne Cruickshank, Thierry Bonnaud, C. Daniel Scott; ‘Progress in Medicinal Chemistry’, Volume 60, 2021, 345-442.
J. H. ter Horst, M. A. Deij, P. W. Cains; ‘Crystal Growth & Design’, Vol. 9, No. 3, 200