Improve and accelerate your crystallization screens
Introduction
New compounds are being produced within the food, personal care, dye, agro and pharmaceutical industries. These new compounds are becoming more and more complex, making dosage determination and formulation difficult. In order to obtain the solid-state form with the desired properties, solid-state screening needs to be performed. Polymorph, co-crystal, salt screening, and amorphous solid dispersion are the most common solid-state screening methods of which, polymorph screening is still the most widely applied. The CrystalBreeder and Crystal16 provide the perfect tool to automate the execution of crystallization experiments, improving reproducibility while drastically increasing productivity and efficiency without compromising on flexibility.
Crystallization screening
Crystallization screens can be a labor and time-intensive process. Therefore, automating your solid-state screening process enables you to perform the crystallization experiments with just a small amount of effort. One Crystal16 can run up to 16 experiments in parallel at a 1-mL scale. The CrystalBreeder can run up to 32 experiments in parallel at a 0.1-mL scale. Reproducibility and controllability enable you to perform good science and meet regulatory requirements.
The solubility of your compound in organic solvents or water plays an important role when designing a crystallization screen. If you want to do cooling crystallization you need high solubility at high temperatures and low solubility at low temperatures. However, if you want to do slurrying with temperature cycling you need to have low solubility at all temperatures. Knowing the solubility will allow you to select suitable solvents for the different crystallization methods. In addition, a selection of solvents and mixtures that spans the range of possible chemical functionalities will maximize the chances of finding new, interesting, and developable solid forms. Both CrystalBreeder and Crystal16 are the perfect tools for determining solubility in an easy and efficient manner.
Setting up your crystallization screen
Performing your polymorph screen using the CrystalBreeder or Crystal16 enables control over the temperature, cooling/heating rate, stirring rate, concentration, and solvent. Additionally, the CrystalBreeder allows you to perform evaporation crystallization or cooling evaporation experiments at a controlled evaporation rate. This enables you to apply a wide variety of conditions including the most commonly used crystallization methods including evaporation, vapor diffusion, and temperature cycling.
A typical polymorph/crystallization screen of 32 experiments performed on a CrystalBreeder would run by using 5 crystallization methods.

Figure 1 Potential Crystalbreeder crystallization screen
A typical polymorph/crystallization screen on the Crystal16 would include 16 experiments by using 3 crystallization methods: cooling crystallization, slurry and temperature cycling.

Figure 2 Potential Crystal16 crystallization screen
Four different solvents could be chosen for the crystallization experiments. Two different cooling profiles could be applied for cooling crystallization (e.g. a fast and a slow cooling rate of 5 °C/min and 0.1 °C/min). Additionally, one can also choose 2 ripening times (short ripening to capture unstable or kinetically stabilized forms and long ripening to allow more stable forms to appear). Different concentrations and different crystallization temperatures can also be considered while designing the crystallization experiments. To fully characterize the solid forms obtained, screening experiments executed on the Crystal16 and CrystalBreeder are typically followed by analytical work using X-ray diffraction, microscopy, spectroscopic techniques, and thermal analysis.
Polymorphic screen example
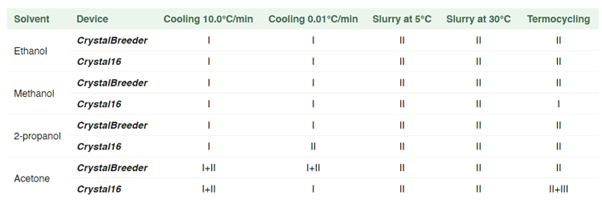
A crystallization/polymorph screening was conducted at Radboud University Nijmegen on isoxazolone dye. The compound is used as filter dye in photographic films and is known to have two polymorphs: needles (form I), and rhombic blocks (form II)1,2. The screen was performed on both CrystalBreeder and Crystal16 systems in parallel.
For this screen, four solvents were used as crystallization solvents. The choice of solvents was made considering the solubility information of the compound in a larger set of solvents. The investigated crystallization methods were cooling crystallization with 10.0°C/min and 0.01°C/min, slurry at 5°C and 30°C for 2 weeks, and temperature cycling.
Besides the already known form I (needles) and form II (blocks), a third form (form III) was also identified.
The resulting crystals of the isoxazolone dye were characterized by X-ray powder diffraction (XRPD) and differential scanning calorimetry (DSC). In addition, scanning electron microscopy (SEM) and optical microscopy images were taken to provide morphological information.

Figure 3 SEM and optical microscopy images of isoxazolone Form I and Form II
Conclusion
The CrystalBreeder and Crystal16 have proven to be suitable for polymorph screening at 0.1 and 1 mL scale. A wide variety of parameters can be varied to perform the desired crystallization methods in a reliable, repeatable, and controllable manner. Allowing you to fast-track molecule development.
Acknowledgments
Technobis would like to thank Evelien Huijs and Dr. Hugo Meekes Radboud University Nijmegen, Solid State Chemistry Department (The Netherlands) for the study and pictures used.
References
1. Aret E., Meekes H., Vlieg E. & Deroover G. Polymorphic behavior of a yellow isoxazolone dye. Dyes and Pigments 72, 339-344, (2007).
2. Aret E., Growth of organic dye crystals: morphology and polymorphism, in Solid State Chemistry, 128, (2013