Salt screening agrochemical with medium through put platform
Introduction
There are several solid forms that are available for development. Each of these solid forms can have vastly different physiochemical properties. These properties mainly fall into two categories: economic and biological. Economic properties such as filterability and flowability determine how easy a solid is to manufacture and process. Biological factors such as stability, melting point, and permeability determine how well an active performs in the field. Economic properties such as morphology, compressibility, and flowability, determine how easy a solid is to handle and produce. Ultimately, the decision on which solid form to take forward for development is based on balancing these factors as well as understanding the thermodynamic relationship between them.
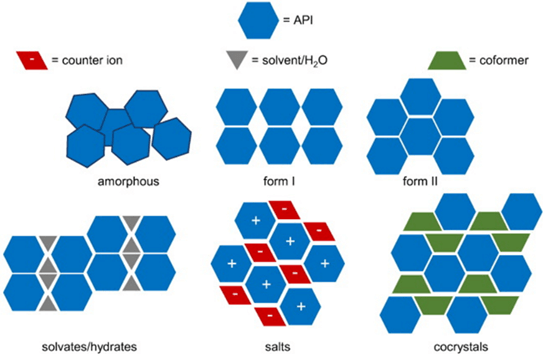
Figure 1 Different types of solid form [1]
Secondary molecules can also be incorporated into crystal structures, and these are hydrates/solvates, salts and co-crystals. Hydrates and solvates are defined when water or a solvent is bonded with the active in a crystal structure. Salts and co-crystals are very similar in structure, where a non-solvent secondary molecule is bonded directly with the active. If the bonding between molecules is hydrogen bonding, then a co-crystal is formed. If the bonding between the two molecules is ionic, then a salt is formed.
Salts are commonly employed in the agrochemical sector to look for new properties that are not seen in polymorphs, as well as extend patient life. The use of salts in agrochemicals has been able to positively change the properties of active compounds, including reducing volatility, improving leaf permeability, and modulating solubility. Additionally, a growing problem within agrochemicals is resistance to actives; this can be mitigated through modifications to the solid form. The use of additional solid forms can also be used to extend the patent life of an active 2,4-D
2,4-dichlorophenoxy acetic acid (2,4-D) is a selective systemic herbicide that was introduced in1950[ii] for use in broad-leaved weeds such as barley, wheat and oats. In 2005 the estimated use of 2,4-D in is various forms was estimated to be 46 million pounds[iii]. The salt form is used to be absorbed by the plant roots. The most common salts of 2,4-D used in industry are the sodium, diethylamine and choline salt.
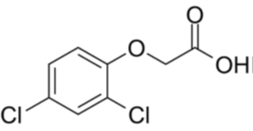
Figure 2 2,4-D chemical structure
Solubility and Salt screening
When designing a solid form screen, understanding of the solubility of the starting material is required in order to use appropriate solvents for every technique. Furthermore, the miscibility of the counter-ion source needs to be considered. Many common counter-ions are introduced by dissolving in water, which is immiscible with common antisolvents like heptane. Therefore, neat solvent and solvent anti-solvent solubilities were collected. Four concentrations were made for each solvent system. Samples were then heated to 5 °C below the boiling point of the solvent (e.g. Ethyl acetate 72 °C) at 0.5 °C/min. Samples were then cooled to 10 °C at 0.5 °C/min. This cycle was repeated 3 times. Solubility points were determined by when the transmissivity reached 100 % (Clear point) and metastable zone limit was determined by when transmissivity dropped to 99 % (Cloud point).
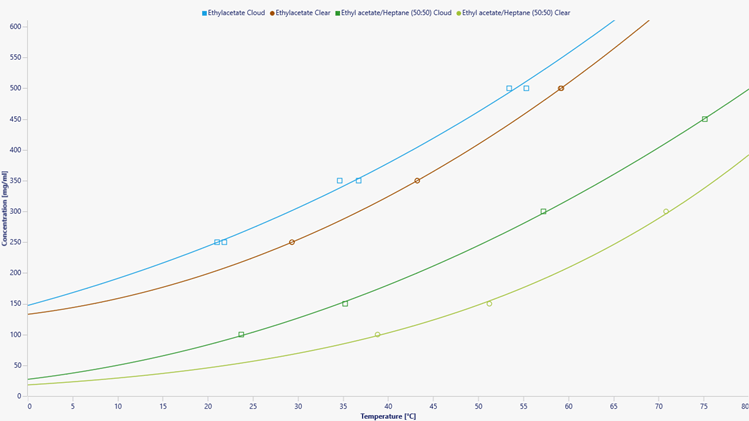
Figure 3 Solubility curve of 2,4 – solvent/antisolvent mixtures
Solubility curves for 2,4-D in several solvent and solvent antisolvent systems were collected. Using the solubility data salt screens using NaOH aq, Chlorine Hydroxide aq and Dimethylamine (DEA) as counter ions were designed in EtOAc and EtOAC/heptane mixture.
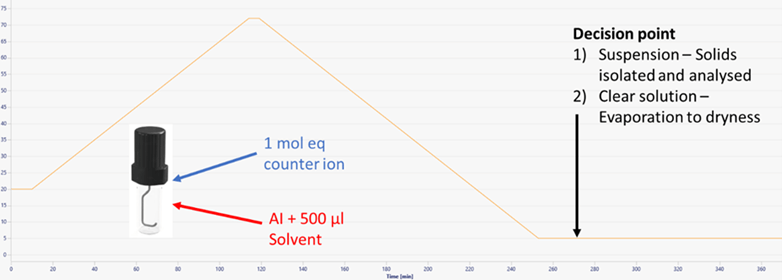
Figure 4 Example experimental profile
Initial experiments were conducted by placing active, 1 mol eq of counter-ion and 0.5 ml solvent in a HPLC vial with a magnetic stir bar. Samples were heated at 0.5 °C/min to 5 °C below the boiling point of the solvent to avoid evaporation. The samples were then cooled to 10 °C at 0.5 °C/min. Any solids produced by cooling were isolated by filtration. Clear samples were then evaporated by removing the cap. All solids were analyzed by XRPD confirming the formation of the salt forms.
Salts
Using the information from the solubility curve, the 3 salts were successfully formed and confirmed by XRPD. DEA salt was also characterized by DSC. The thermogram of 2,4-D shows a melting point endotherm with an onset of 138 °C, whist the new DEA 2,4-D salt showed a lower melting point endotherm with an onset 87 °C. Whilst this is lower than the free acid it would still make it suitable for using in suspension concentrate formulations.
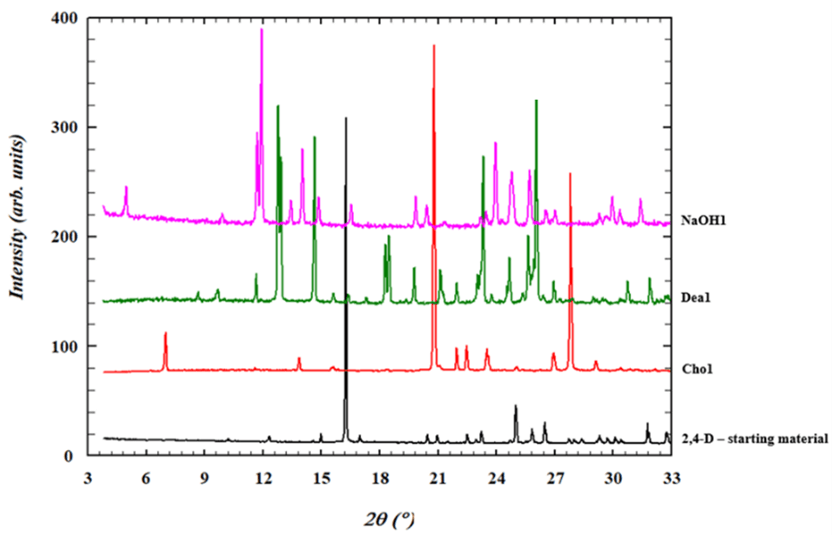
Figure 5 XRPD overlay of 2,4-D, Choline salt, Diethyl amine salt and sodium salt.
It was observed that the Choline (Cho) salt generated began to deliquescence at room humidity. This would make this salt unsuitable for use in a formulation such as suspension concentrate but could be used as an emulsion concentrate.
Conclusions
We demonstrated how the Crystal16 can be used as a screening platform for salts of the commonly used agrochemical 2,4-D. Initially solubility curves for the active were collected in several solvents and solvent/antisolvents to find suitable solvents and concentrations. Three counter ions were used, NaOH, DEA and Chol to successfully generate commercially used 2,4-D salts utilizing controlled cooling and evaporation. The successful formation was confirmed with XRPD analysis.
The Crystal16 is an ideal multi-well platform for medium throughput screening. Four independent temperature-controlled zones allow you to screen up to 16 samples at one time. It is possible to rapidly screen solvents, counter-ions and temperature and stirring conditions for identifying new salts.
Acknowledgments
Technobis Crystallization Systems team is deeply thankful to Monica David, Claudia Brăilă, Mihaela Pop and TeraCrystal for their help with this application note.
References
[i] Paolo Cerreia, Vioglio , R.Chierotti Roberto Gobetto; Advanced Drug Delivery Reviews Volume 117, 1 August 2017, Pages 86-110
[ii] http://sitem.herts.ac.uk/aeru/ppdb/en/Reports/4.htm
[iii] https://archive.epa.gov/pesticides/reregistration/web/html/24d_fs.html

Crystal16