Why predicting solubility is important
Solution crystallization is a necessary unit operation for the separation, purification, and crystal modification in active pharmaceutical ingredient (API) production. Solubility is essential information for commencing the crystallization process. For cooling crystallization, the solubility curve describes the relationship between solubility and temperature for a selected solute-solvent system.
For the discovery of high-end drugs, it becomes challenging to know the solubility varying with several parameters (temperature, solvent, solvent composition) considering the shortage of time and material for some crucial APIs. Consequentially, predicting tools comes into play an significant role, as it provides wider information by interpolating/extrapolating the limited experimental data. Additionally, predicted data enables the scientist to choose a suitable solvent with a high solubility for the solute and implicitly estimate the theoretical yield for a crystallization process.
Case Study: Effect of solvent composition on solubility of ascorbic acid
The solvent employed for the crystallization of ascorbic acid significantly affects its solubility. The binar y mixtures of water and alcohol have intermediate solubility depending on the solvent composition. Predicting the solubility of ascorbic acid in water-alcohol solvent mixtures from the Crystal16 experimental data is the objective of this case study.
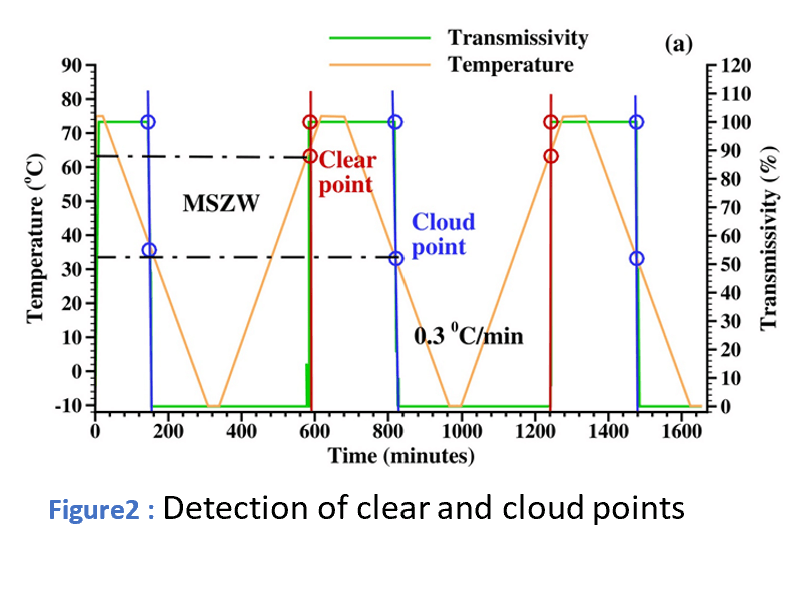
Measuring the solubility with Crystal16
The solubility of ascorbic acid in 16 solvents (12 binary mixtures and 4 pure solvents) was measured by the polythermal method using the Crystal16 instrument.
Results
Ascorbic acid has a high solubility in water, followed by methanol, ethanol, and isopropanol in decreasing order. Solubility is a function of the interaction between solute and solvent. Ascorbic acid is a polar molecule with four hydroxyl groups and two hydrophilic oxygen atoms. Consequently, the solubility of ascorbic acid is high in highly polar solvents like water and progressively lower in alcohols of decreasing polarity.
Modelling solubility using Jouyban-Acree model
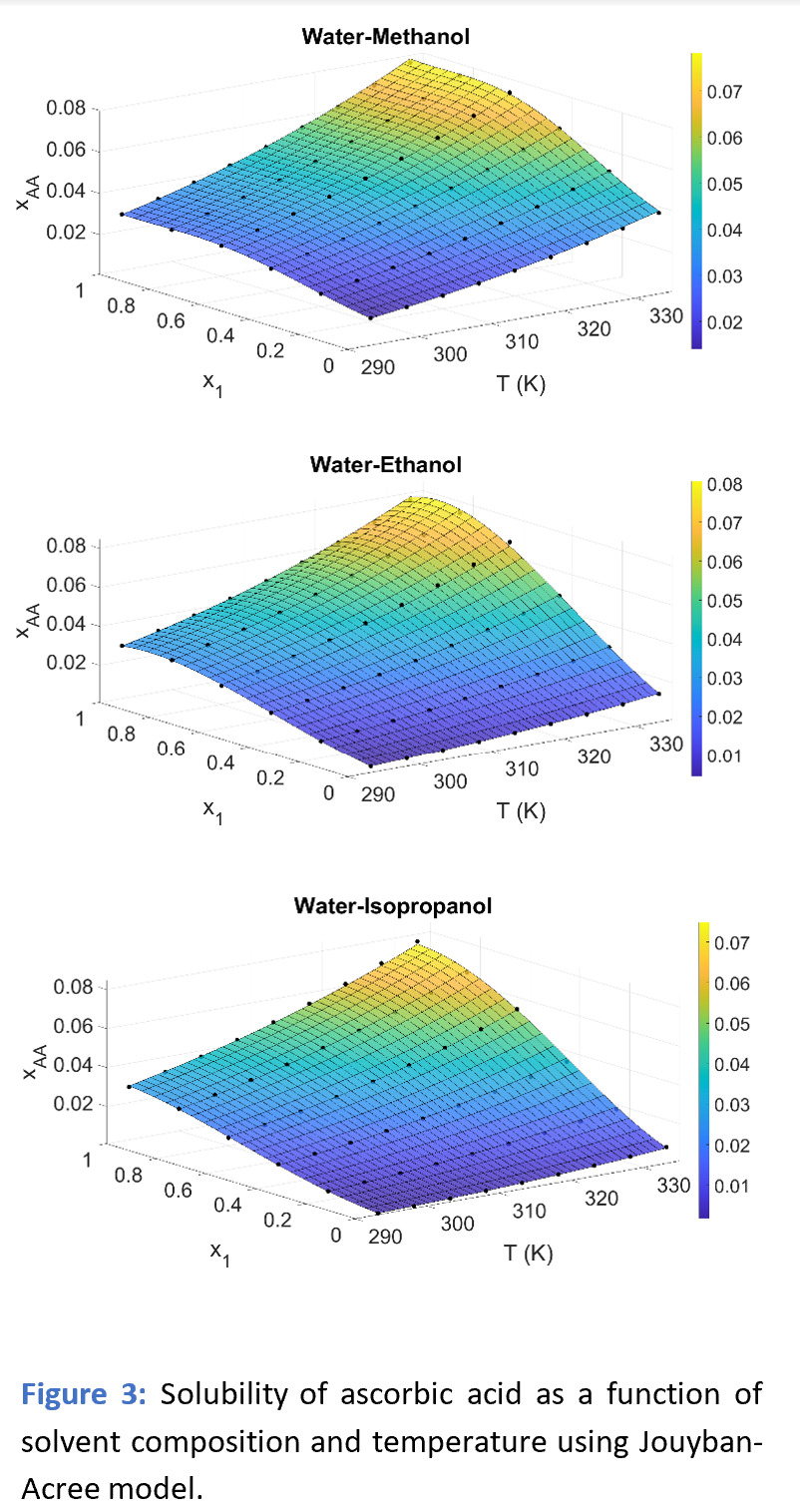
Where xAA is the mole fraction of ascorbic acid in a binary mixture, x1 is the mole fraction of water in a binary mixture. T is the absolute temperature. A1, A2, A3, A4, A5, A6 and A7 are seven model parameters. Using model parameters, the surface plots (Figure below, RHS) show the predicted solubility as a function of temperature and solvent composition simultaneously. The color contour shows the solubility of ascorbic acid in mole fraction (xAA), varying with the temperature and solvent composition. The yellow color at higher temperatures (T) and higher mole fraction of water in binary solvent systems (x1) reflects the high solubility.
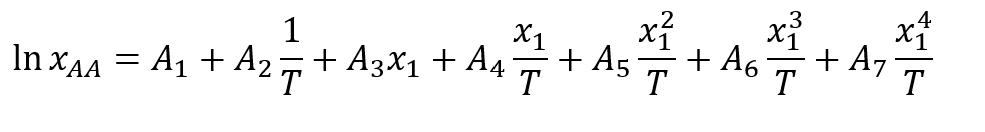
Conclusion
Experimentally measured solubility data using Crystal16 in various water-alcohol solvent systems was accurately correlated using Jouyban–Acree model. The surface plot of the predicted values shows good agreement with the experimental values. The model enables us to predict the solubility of ascorbic acid as a function of: temperature and solvent composition simultaneously. Knowing the solubility in various solvents makes it easier to select the most appropriate solvent to optimize the crystallization process. In addition, predictive technology provides us with a wide range of information with limited experimental data and resources, improving efficiency by reducing cost and time to market for an API.
Curious to learn more?
Read more about this case study in the Application Note: "From Experimentation To Modelling: Understanding The Effect Of Solvent Composition On Solubility".