Why obtaining crystallization data at the right time is crucial
Ritonavir hit the market in 1996 as an antiretroviral agent, part of a seemingly miraculous therapeutic array capable of treating HIV. Just two years later, however, analysis detected a less bioavailable polymorphic form, capable of compromising the effective conformation and as a result, the drug was wiped from the shelves. Poor access to the thermodynamic data caused researchers to miss the ineffective, more stable polymorph and ultimately cost its manufacturer $250 million.
In this article, we explain how innovations in benchtop instruments are delivering crucial data at the earliest stage, to help researchers streamline drug development process.
One step at a time
Getting novel drugs to market is a famously expensive business. In 2021, global expenditure on research and development (R&D) alone by American pharmaceutical companies reached $102 million. The costly development process, which carries no guarantee of success, is often quoted as a major reason for the slowdown in new drug production.
Of course, the development chain is comprised of many steps. Improving the efficiency of any of these steps, however, has the potential to help get medicines to the people who need them quicker. One such step, prime for optimization, is crystallization studies.
Crystallization in the development process
The solid form of an active pharmaceutical ingredient (API) is critical to its material properties, such as solubility and morphology. As such, investigating parameters including particle size distribution (PSD), crystal shape and quality, and the most stable polymorphic form are key elements of crystallization research. Using the resulting data, solid state scientists can map a path of reaction environments and conditions to formulate drugs effectively.
The traditional methods by which these investigations are conducted are limited, especially in the current geopolitical climate. Access to manufactured APIs is scarce and unreliable, and excessive working volumes in laboratories compounds this logistical issue.
Another contributing factor to drug development inefficiency is the underwhelming success rate of drug molecule candidates. Research has found that only 10 to 15% of small-molecule drugs make it from the pre-clinical stage to market. To boost efficiency and spend less on dead-end molecules, researchers need ways of ruling out them out earlier in the process.
Addressing crystallization frustrations
One way to solve both these issues is employing smaller working volumes in the lab. For example, the Crystalline V2 instrument uses 2.5-5 ml of starting material, which is the smallest volume at which scalable PSD information can be collected using transmissivity techniques, is an order of magnitude reduction from the market standard.
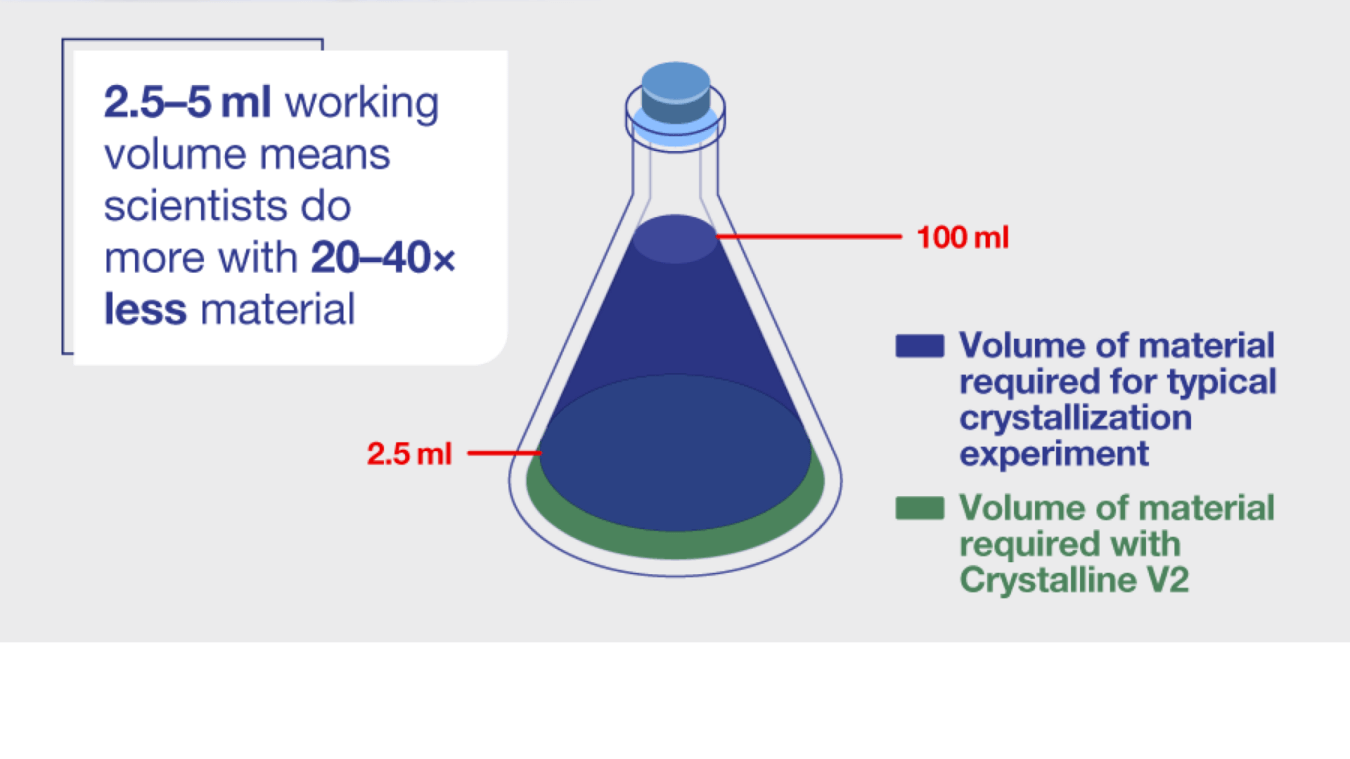
The benefits of this are plain: the potential number of experiments conducted vs. amount of starting material consumed can increase manyfold. Not only does this make the resulting data cheaper and cut down waiting times from API supply chains, but conclusions about a small-molecule drug’s eligibility for further research can be reached more quickly.
A knock-on effect of using smaller working volumes in crystallization experiments is less waste to be disposed of afterwards. Paired with in-line analytics technology — meaning samples are analyzed within sealed, disposable reaction vessels — the time lost to cleaning and resetting apparatus is easily minimized.
Collectively, these factors could allow scientists to run three times as many experiments in the same time. Over the full development cycle, the impact is amplified.

Another recent technological step forward that empowers scientists with data is upgraded visualization hardware, with improved resolution and magnification enhancing the quality of information obtainable from the crystallization process. The former lets the viewer see more detail about the crystal size, morphology and classification. The latter reveals smaller particles, as well as surface quality details of larger particles, like cracks and accumulation points for contaminants, such as mother liquor.
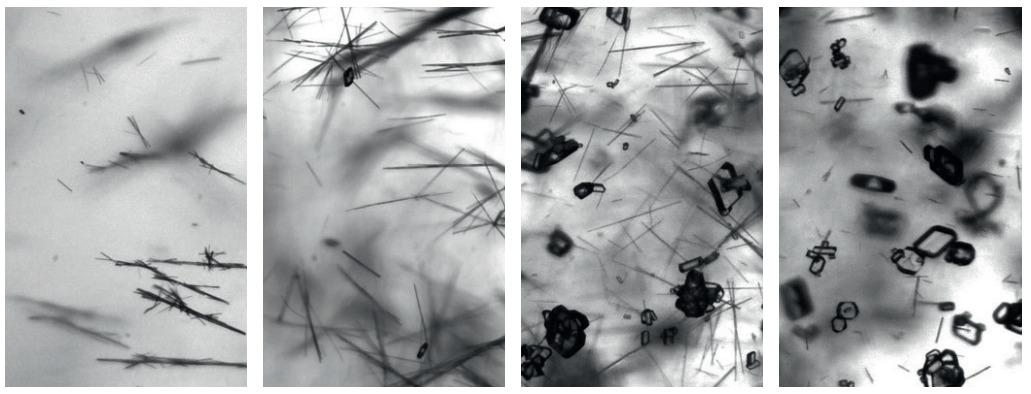
Polymorphic transformation seen with the Crystalline instrument

Detection of different crystal shapes and sizes in the Crystalline instrument using AI-based image analysis software
Some instruments, such as Crystalline, are capable of 0.63 microns per pixel of resolution and 6x magnification. This advanced hardware enables collection of data on close to a nano scale, in real-time as researchers conduct experiments.

Last but not least, having instruments with a small footprint helps save precious lab space. Next-generation platforms have evolved to meet this need.

Conclusion
If novel drug production is to rise to meet the challenges of the 21st century, then the pharmaceutical industry must learn from Ritonavir and the lessons of the past. Timely data collection during the drug development pathway is a step in the right direction.

Curious in the latest innovation?
Read more about Crystalline instrument, a new and unique analytical instrument that allows scientists to access crystallization and formulation information at milliliter scale.