Salt screening with the crystal16: design and considerations
Introduction
More than half of all drug molecules on the market are salts. The main reason behind this is that in general salts have more desirable aqueous solubility and bioavailability profiles compared to the free form of the active pharmaceutical ingredients (API). Additionally, by using salts it is possible to modify chemical and mechanical properties such as stability and manufacturability. The difference between the physio-chemical properties of different salts of the same API can be so great, that it has been said that ‘changing the salt is changing the drug’. This is true in the legal context as well, as new API salts are patentable. Therefore, salt screenings have become increasingly important for protecting your intellectual property, and not only as a way of improving and tuning APIs properties.
Challenges
Strong acids and bases readily form salts with one another. However, pharmaceutical compounds are typically weak acids and bases so can be a challenge to form salts. In these cases, it is recommended that the difference between the pKa of the compound and counter ion is higher or equal to 3 to increase the probability of salt formation. If the difference is between 0 and 3, then the resulting solid form can be either a salt or a cocrystal, depending on each individual chemical system. This is especially true when dealing with weak organic acids or bases, and in such cases, additional analyses are required to confirm if the new solid form is indeed a salt.
While this pKa rule is a good starting point for designing a salt screen, chemists should take into account that most pKa values reported in the literature are for aqueous solutions. The acidity/basicity of any compound is highly solvent dependent, and the same experiment performed in water and in isopropanol can yield different results, and salts can be missed as protonation has not been fully completed. Furthermore, the counterions available are limited to those found on the Generally Regarded As Safe (GRAS) list when dealing with pharmaceutical compounds.

Figure 1 Salt-cocrystal continuum as a function of ΔpKa
The current state of salt screening experiments has moved away from high-throughput screening, towards a more focused medium-throughput approach relying on a rational design of experiments. The pKa rule and API and counterion solubilities are used as starting points for designing a rational salt screening DoE. In practice, this translates to testing several counter-ions in several solvents.
We present here some common salt screening methodologies which are described in the literature. All rely on the mixing stoichiometric amounts of API and counter ion and utilize either solution or slurrying-based techniques. All these methods are possible using the Crystal16 instrument allowing for reliable control of temperature and agitation than performing the same experiments by hand.
Methods
In-Situ salt screening
In this method, an amount of the free-form API and a stoichiometric amount of acid or base are slurried with stirring for several days using the Crystal16 instrument isothermally or with temperature cycling. The solids are then isolated by centrifugation or filtration and analyzed to confirm the solid form. This method is useful in cases where the free-form API shows low solubility.
This strategy can be applied using the Crystal16 by simultaneously testing 16 different acids or bases, while accurately controlling the temperature, the stirring rate, and monitoring any potential dissolution events by making use of the transmissivity technology. The Crystal16 combines automation with integrated turbidity measurement to determine cloud and clear points and is ideally suited to acquire crystallization-related data at an early stage using only minimal amounts of sample.

Figure 2 In-situ Salt Screening diagram
Saturated solution method
This method is performed by dissolving free-form API in a solvent and then adding a stoichiometric amount of acid/base. The solution is aged or cooled with any precipitate isolated and analyzed to confirm the solid form. A key piece of information used in this approach is the solubility of your API in several solvents, as well as that of each counterion. This approach has the benefit of covering a very large chemical space, testing several counterions in several solvents at the same time. This of course translates into potentially more solid forms discoverable in the screening.
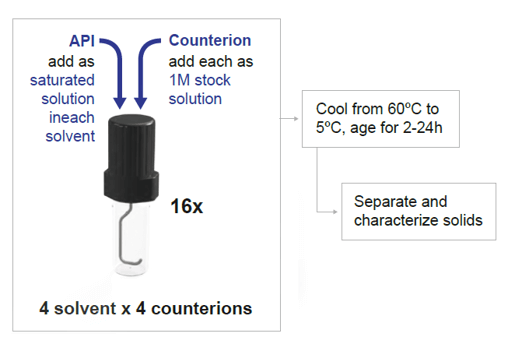
Figure 3 Saturated Solution Method diagram
The Crystal16 is ideally suited for such a combinatorial study: the device allows for the monitoring of 16 individual crystallization reactors, arranged in a 4x4 matrix. This means testing 4 different counterions in 4 different solvents at the same time at your desired heating and cooling rate.
Cooling evaporative method
In this last method, stoichiometric amounts of API and acid/base are dosed as solids in each vial, to which an appropriate amount of solvent is added. The resulting slurries can be aged at different temperatures or thermocycled. If solids are not observed, the solutions are subsequently evaporated.
The main advantage of using the Crystal16 for such screening is the ability to track any dissolution or crystallization events with temperature in real-time.
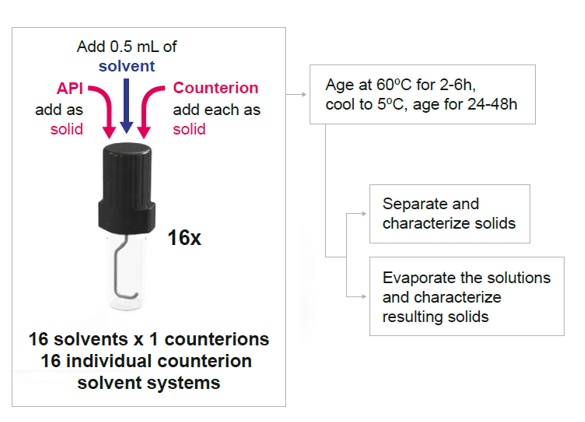
Figure 4 Cooling Evaporative Method diagram
Conclusions
Salt screening is more than mixing your API and counterion together. Although apparently simple, in a salt formation reaction one should consider not only the API properties but also those of the counterion, the solvent involved, together with many other experimental conditions, such as temperature and stirring rate. Several salt screening methodologies can help to cover as much of the chemical space as possible, and using rational approaches also reduces the number of experiments that need to be performed. In any case, the Crystal16 device, having 4 individual temperature and stir-controlled zones, and 16 reactors each with individual in-line transmissivity analytics, is ideally suited to help you navigate the solid form salt landscape of any API.
References
Fernández Casares, A., Nap, W.M., Ten Figás, G., Huizenga, P., Groot, R. and Hoffmann, M. (2015), An evaluation of salt screening methodologies. J Pharm Pharmacol, 67: 812-822. https://doi.org/10.1111/jphp.12377
Paolo Cerreia Vioglio, Michele R. Chierotti, Roberto Gobetto (2017), Pharmaceutical aspects of salt and cocrystal forms of APIs and characterization challenges, Advanced Drug Delivery Reviews, 117, 7, 86-110. https://doi.org/10.1016/j.addr.2017.07.001
Benjamin M. Collman, Jonathan M. Miller, Christopher Seadeek, Julie A. Stambek & Anthony C. Blackburn (2013) Comparison of a rational vs. high throughput approach for rapid salt screening and selection, Drug Development and Industrial Pharmacy, 39:1, 29-38. http://dx.doi.org/10.3109/03639045.2012.656272
Jun Qiu, Anisha Patel, Jason M. Stevens (2020) High-Throughput Salt Screening of Synthetic Intermediates: Effects of Solvents, Counterions, and Counterion Solubility, Organic Process Research & Development 2020 24 (7), 1262-1270. https://doi.org/10.1021/acs.oprd.0c00132