Many large, medium, small size pharma companies around the world are racing to find a cure and/or a vaccine for COVID-19. Several companies are teaming up to research older drugs earlier developed for other purposes. At certain extent, this race resembles the one for ritonavir in the late 90’s, and the race to screen as efficient and comprehensive as possible to understand the polymorphic behaviour of the drug. For those that do not know the story of Ritonavir, the drug in crystal form I was introduced on the market in 1996. In 1998, a lower free energy, more stable polymorph, crystal form II, was obtained. Due to its lower oral bioavailability, the oral capsule formulation had to be temporarly removed from the market and meanwhile to extensively screen and understand the polymorphic behaviour of the drug.
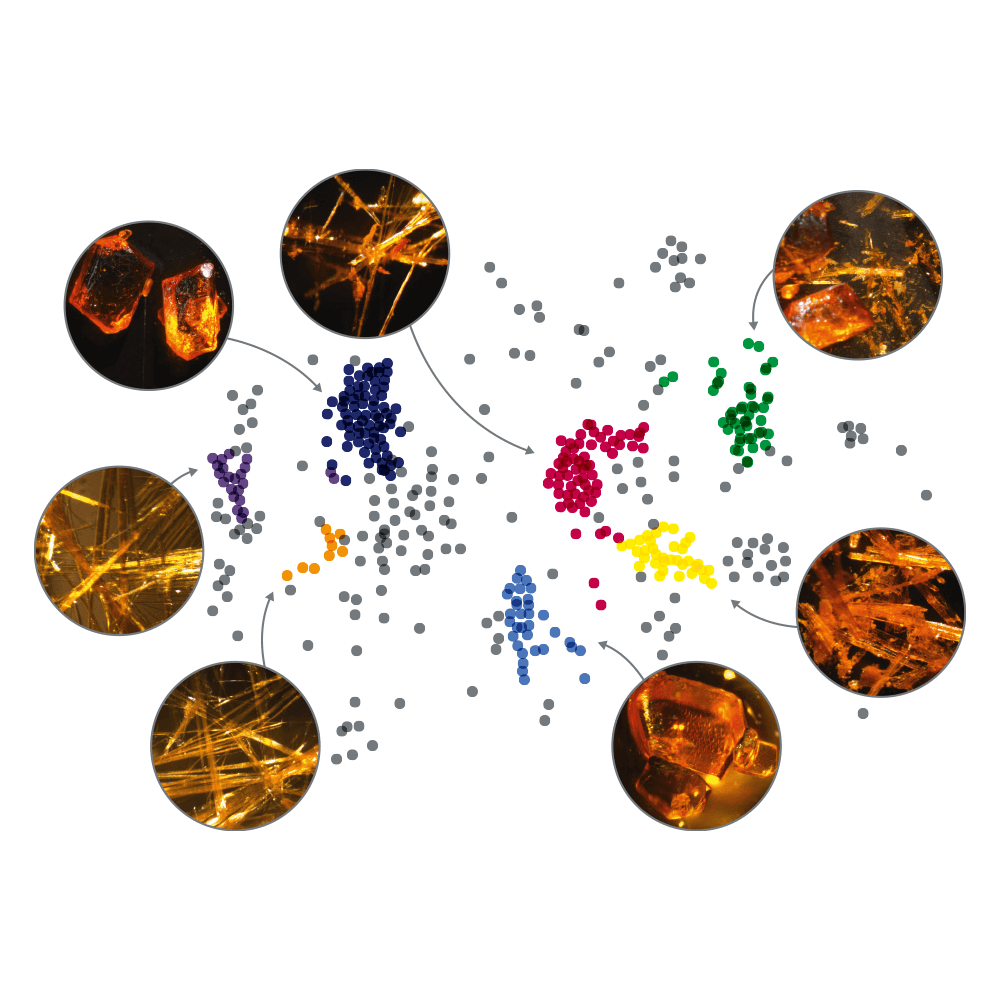
Finding the optimal form with the best characteristics for development is critical. When the compound reaches the batch-production stage, reproducibility of the desired polymorphic or salt form is of key importance in the development of the compound.
To be able to select the optimal form, knowledge of as many as possible forms is needed. Crystallization is the experimental technique used to perform solid form screens. Understanding the solid state of your active ingredient will lead you to full properties acquirement. Polymorphs, as well as solvates or hydrates, salts or cocrystals of your active ingredient may differ in key properties such as solubility, dissolution rate, stability, and particle habit.
Technobis Crystallization Systems' workflow - consisting of 3 instruments - are the perfect tools for crystallization screening at different working volumes. Depending on the amount of material available, now scientists around the world can perform solid form screens at microliter scale with the CrystalBreeder, at 1mL scale with the Crystal16 and at 5mL scale with the Crystalline instruments.

Technobis Crystallization Systems Workflow
The crystallization space can be systematically explored with the ability to control temperature profiles, stirring speed and crystallization mode. Whether you are searching for new polymorphs, or doing salt or co-crystal screens, the transmission technology gives you a clear indication of a hit.
Several crystallization methods are now available:
- Cooling crystallization
- Evaporation crystallization
- Combined cooling evaporation crystallization
- Vapor diffusion crystallization
- Slurry
- Thermocycling
- Anti-solvent crystallization
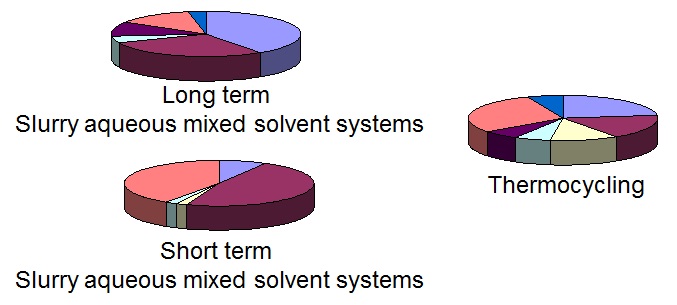
The Crystal16 offers invaluable assistance throughout the various stages of the drug development life cycle. Medium throughput screening can be performed on the Crystal16. Sistla et al from Pfizer described in 2011, a medium-throughput screen using the Crystal16 with an accelerated thermal cycle in a mixed solvent system. They have successfully tested nine compounds to demonstrate:
- the utility of the method
- use of Crystal16 in hydrate screening
- a preferred cycle and
- solvent system were identified.
With only 150-250 mg of compound a medium throughput screen can be performed by using the either the CrystalBreeder or Crystal16 instruments.
Additionally, the Crystal16 provides an accurate temperature control and in combination with the transmission technology or the Crystalline PV with the camera technology, both offer scientists the perfect tool to easily assess solubilization.
Last but not least, as McCrone said long time ago, "the number of forms known for a given compound is proportional to the time and money spent in research on that compound". In hope that time and money spent in your laboratories can be reduced, Technobis Crystallization Systems' Workflow brings now to you the aiding tools to your crystallization screens.
REFERENCES
- Sistla A., Wu Y., Khamphavong P. Liu J., Medium-throughput hydrate screening using the Crystal 16™, Pharmaceutical Development and Technology, 2011, 16(2), 102–109.
- P. Láng, V. Kiss, R. Ambrus, G. Farkas, P. Szabó-Révész, Z. Aigner, E. Várkonyi, Polymorph screening of an active material, Journal of Pharmaceutical and Biomedical Analysis, Volume 84, 2013, Pages 177-183,ISSN 0731-7085.
- P. Láng, E. Várkonyi, J. Ulrich, P. Szabó-Révész, Z. Aigner, Analysis of the polymorph changes of a drug candidate, Journal of Pharmaceutical and Biomedical Analysis, Volume 102, 2015,Pages 229-235,ISSN 0731-7085.
- Masatoshi Karashima, Kouya Kimoto, Takashi Kojima, Yukihiro Ikeda, Rational polymorph screening based on slow cooling crystallization of poorly soluble mebendazole, Journal of Crystal Growth, Volume 390, 2014, Pages 30-37, ISSN 0022-0248.
- Ana Fernández Casares, W. Mieke Nap, Glòria Ten Figás, Pieter Huizenga, Richard Groot and Marcel Hoffmann, An evaluation of salt screening methodologies, Jounal Pharmacy and Pharmacology, 2015, 67(6):812-822.
Curious to learn more?
Discover our Workflow – CrystalBreeder, Crystal16 and Crystalline instruments – for fully controlled crystallization studies.