Biotherapeutics
Biotherapeutics have seen a significant increase in use, approval and development in recent years. Each year the number of approved new biotherapeutic treatments is increasing with half of those being monoclonal antibodies, most notably in the vaccine for Covid19. However, the manufacturing costs for biotherapeutics are significantly higher than traditional small molecule actives. One reason for the higher costs producing and manufacturing biotherapeutics is the isolation process. Isolation is typically done by chromatography with up to 80 % using the more expensive affinity chromatography to isolate products which can results in a treatment cost in the tens of thousands of pounds.
The isolation of small organic molecules is typically done by crystallization which is a very common and cost effective process. This is not always possible with biotherapeutics due to the large molecular size and flexibility; coupled with the complexity of multicomponent mixtures making crystallization difficult. Porous silica particles are commonly used as templating particles to induce nucleation as pore size has been shown to be an important factor in the effectiveness of the templating particles to induce nucleation with the pore size needing to be 2-10 times the radius of gyration to stabilize the nucleus. Therefore, well designed template particles can offer a way for isolating by crystallization of large molecules reducing the cost of production and manufacturing.
An additional hurdle to crystallization development of biotherapeutics is the availability of material. This can limit the number of experiments that can be done to find conditions crystallization can occur. With an intuitive software interface, the CrystalBreeder controls and analyzes real time turbidity information on 32 reactors, within 8 independent temperature zones, using as little as 1 mg of compound at working volumes of 0.06 - 0.1 mL. The CrystalBreeder offers the unique ability to rapidly generate solubility information, screen crystallization conditions and calculate nucleation rates at microlite scale reducing the amount of material needed.
Nucleation rate and induction time
Crystallization consists of two main processes, nucleation and growth. A method for determining nucleation rate from a series of induction time measurements. Initially discussed by Turbull[i] and was later expanded on by Myerson[ii], it was then applied to the Crystal16 instrument utilizing the transmissivity data by ter horst. [iii] By measuring the induction time, which is defined as the time between supersaturation generation and crystallization (cloud point) it is possible to plot as probability of crystallization against time.
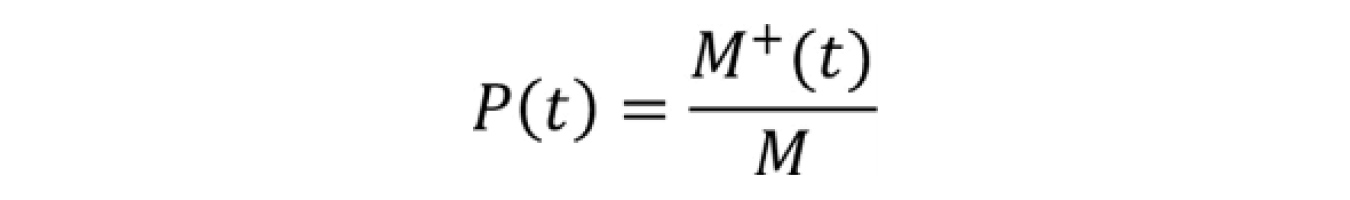
Where P(t) is the probability of a vial crystallizing by time t, and M+(t) is the number of vials crystalized by time t and M is the total number of vials tested in that data set.
The induction time data can be fitted with probability function against time graph and fitted with the equation below. From this fitting the nucleation rate (J) can be estimated as the initial slope of the fitted curve.
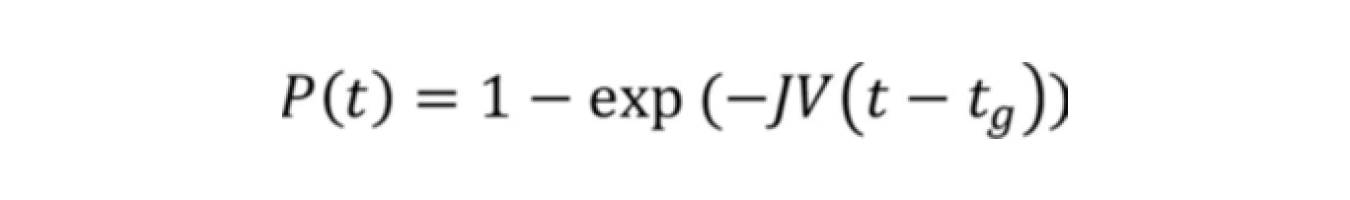
Where P(t) = probability of nucleation at time (t), J = nucleation rate, V = volume, tg = the time from nucleation until detection (growth time
Experimental design
Monoclonal antibody Anti-CD20 stock of concnetration9 mg/ml in a stock buffer (25 mM sodium citrate, pH = 6.5, 154 mM NaCl) were stored at -80 C in 1 ml vials until used. Samples were thawed and the stock buffer was exchanged using centrifugation filter to the crystallization buffer 100 mM HEPES, pH = 7.4, NaOH solution). Final concentration of Anti-CD20 was determined to be 100 mg/ml by UV-Vis spectroscopy.
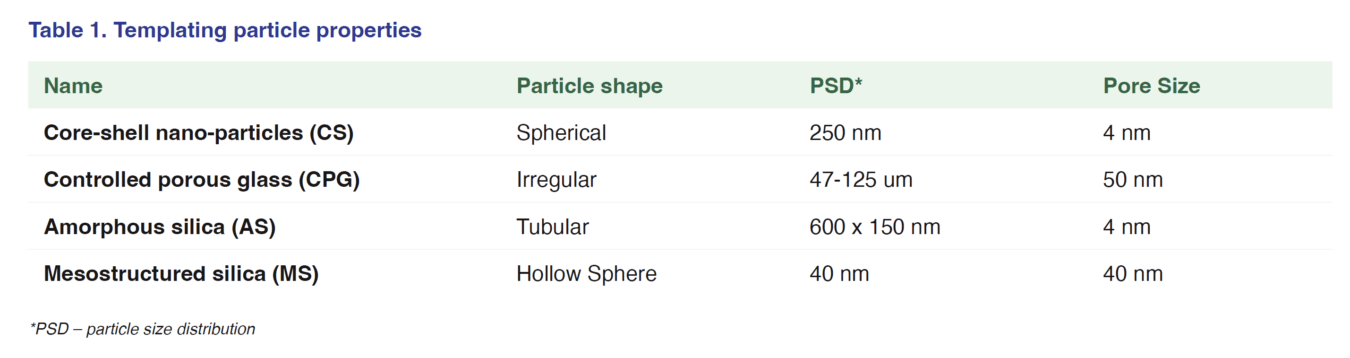
Templating particles were prepared in a salt solution. The salt solution made by dissolving PEG-400 and Na2SO4 in the crystallization buffer giving a final template particle concentration was calculated to be 0.5 mg/ml which is below the limit of detection.
Crystallization conditions were chosen as 25 mg/mL of Anti- CD20, 0.75 M of Na2SO4, 7.5% of PEG- 400 (resulting in a supersaturation of around S = 1.25) in the presence or absence of 0.5 mg/mL of template, at 20°C under stirring conditions (700 rpm). Crystallization was noted as when transmissivity first dropped below 100%.
Results
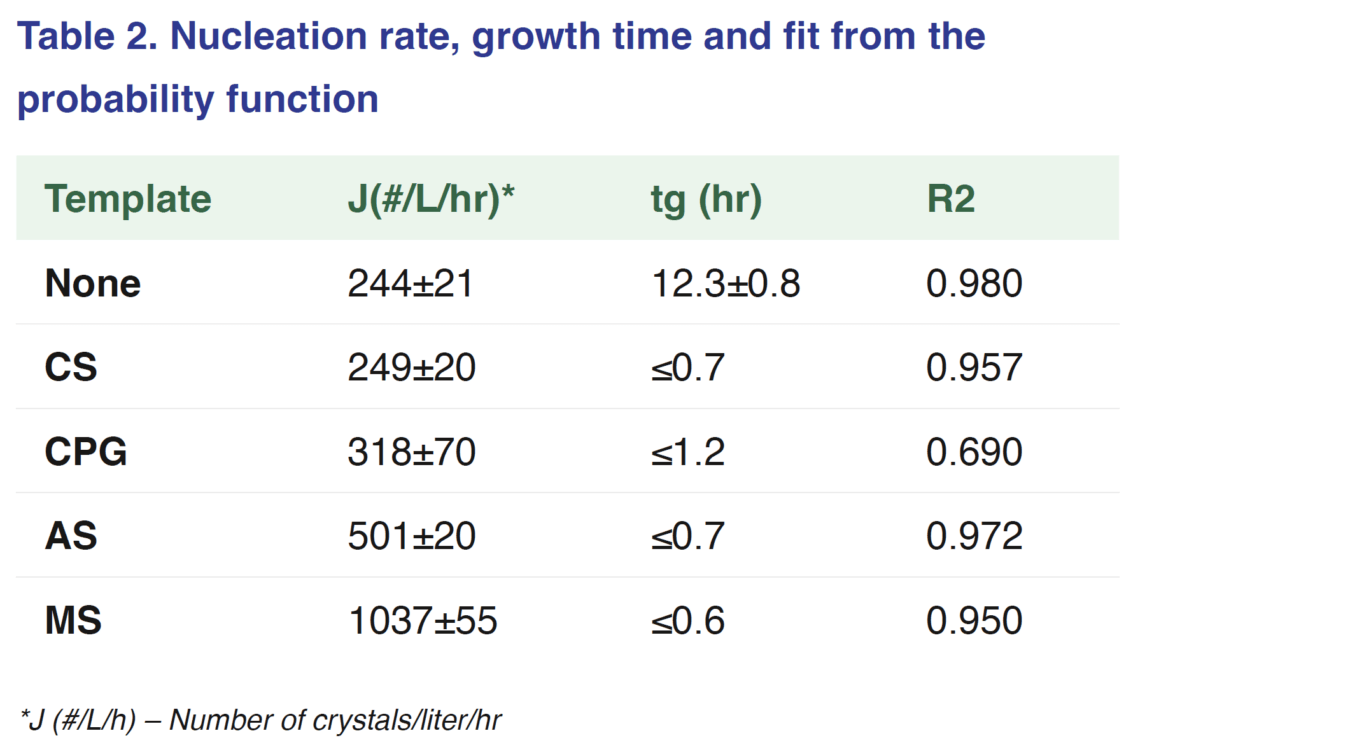
The results of the induction time experiments show there is a reduction when templating particles are present with the median induction times for templates being 2 hrs and without templates 26 hrs.
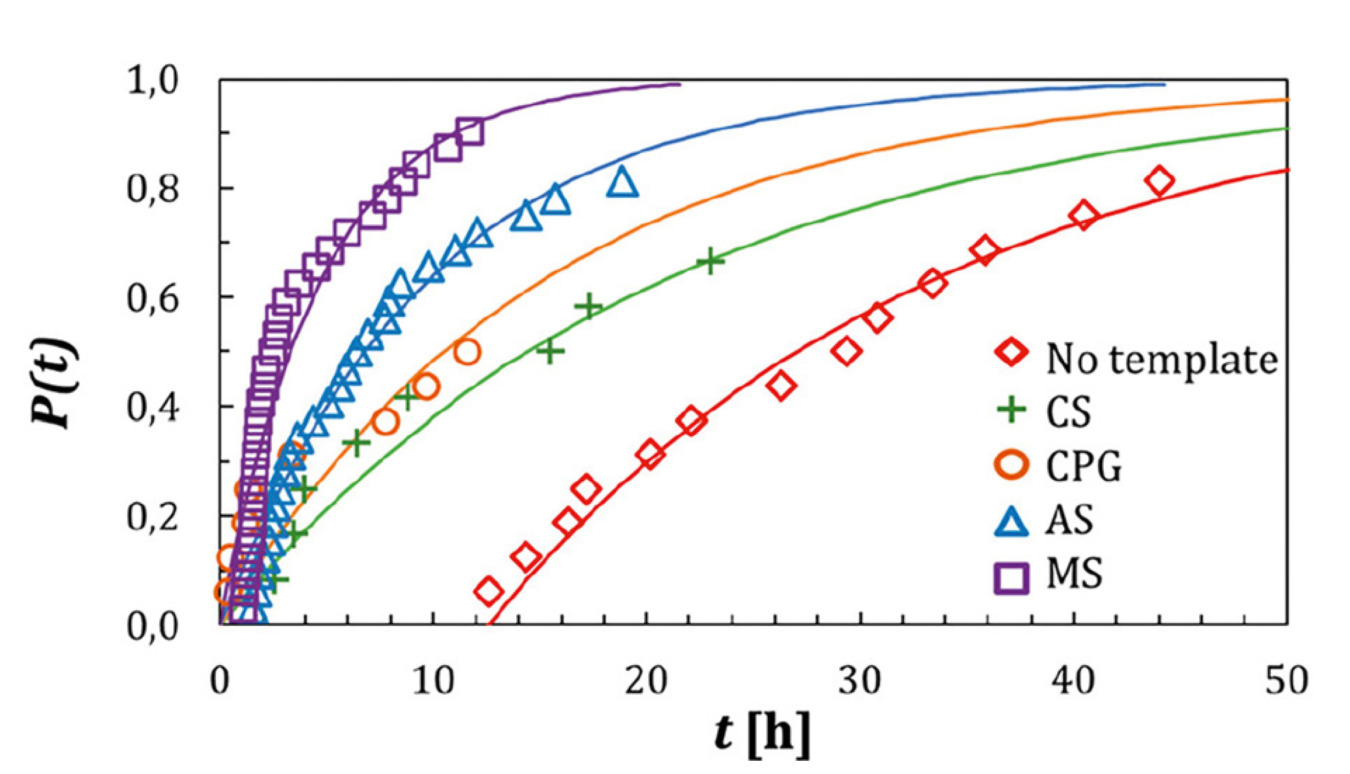
Figure 1. Nucleation probability graph against time
This is also reflected in the tg where there is a reduction from 42 to 12.3 hrs with the addition of template particles. However, this decrease in induction time and tg was not reflected in an increase in the nucleation rate in all cases. Significant increases in the nucleation rate were seen when AS and MS template particles were introduced increasing to 501 and 1037 #/L/hr respectively. This can be explained by the difference in pore size, where if the pores are large enough to trap molecules, but also small enough to stabilize the nuclei. Pore shape can also explain the increase slight increase seen in AS particle dispute the pore size being small.
Conclusions
Presented here is method for investigating the effect templating particles have on the nucleation rate of the biotherapeutic Anti-CD20. Whilst the presence of templating particles reduced the induction time, this did not translate to an increase in nucleation rate. Significant increases were seen in amorphous silica and mesostructured silica. Using the CrystalBreeder easy to use software with inline transmissivity analytics it possible to measure nucleation rates of different conditions at microliter scale when material is limited. This results in more experiments that can be conducted to find condition crystallization can occur in hard to crystallize systems like biotherapeutics.
Acknowledgements
TCS would like to thank Prof. ter Horst, Charline J. J. Gerard, Maria L. Briuglia, Nazer Rajoub, Teresa F. Mastropietro, Wenqian Chen, Jerry Y. Y. Heng, Gianluca Di Profio, CMAC and the University of Rouen for help with the application note.
[1] Turnbull, David (1952). Kinetics of Solidification of Supercooled Liquid Mercury Droplets. The Journal of Chemical Physics, 20(3), 411–. doi:10.1063/1.1700435
[2] Knezic, Dragutin; Zaccaro, Julien; Myerson, Allan S. (2004). Nucleation Induction Time in Levitated Droplets. The Journal of Physical Chemistry B, 108(30), 10672–10677. doi:10.1021/jp049586s
[3] Jiang. S; and ter Horst. J, Crystal Nucleation Rates from Probability Distributions of Induction Times., Cryst. Growth Des. 2011, 11, 1, 256–261, https://doi.org/10.1021/cg101213q
Curious to learn more?
Read more about this case study in the application note "Crystallization Of Antibodies With Templating Particles".