Challenges to measuring nucleation rates
Crystal nucleation is one of two key processes in the formation of crystals, with the other being growth; and is a key part in deciding important particle properties such as polymorphism, morphology, and particle size distribution. Therefore, understanding the underlying mechanism, and kinetics can allow for particle properties to be optimized. However, Measurement of primary homogenous nucleation is a difficult and time-consuming process made more complex by the stochastic (random) nature of nucleation; therefore, it is imperative that temperature and thus supersaturation is tightly controlled. An additional hurdle to accurately measure nucleation rates is that crystal nucleus are generally 1-1000 molecules in size and cannot currently be observed using available analytics. Further challenges to measuring nucleation rates are seen in additional processes that are happening alongside nucleation such as growth, agglomeration, and secondary nucleation which can distort the measured nucleation rate.
A method to approach these challenges
A method was developed utilizing the Crystal16’s transmissivity analytics coupled with the first commercially available software for measuring nucleation rate to address these challenges, though measuring the variation in induction times in identical stirred solution at small scale. With this method, nucleation rates have been successfully demonstrated in multiple systems in a short period of time, which can be further reduced by making use of the Crystal16’s feedback control software feature.
Induction time measurements
Induction time is defined as the time between supersaturation being generated and the first crystal appearing. Observations linking the multiple induction time of nucleation and probability of nucleation were discussed by Turnbull[i] whilst looking at the solidification of mercury droplets. This was expanded by Myerson et al[ii] while investigating homogeneous nucleation in levitated drops of the protein lysozyme showing it is possible to calculating the nucleation rate from a large number of identically small-scale experiments looking at the induction time of crystallization and the number of droplets that crystallize in a given time. The application of calculating the nucleation rate from induction time to an accessible scale using the Crystal16 was done by ter Horst and Jiang.[iii]
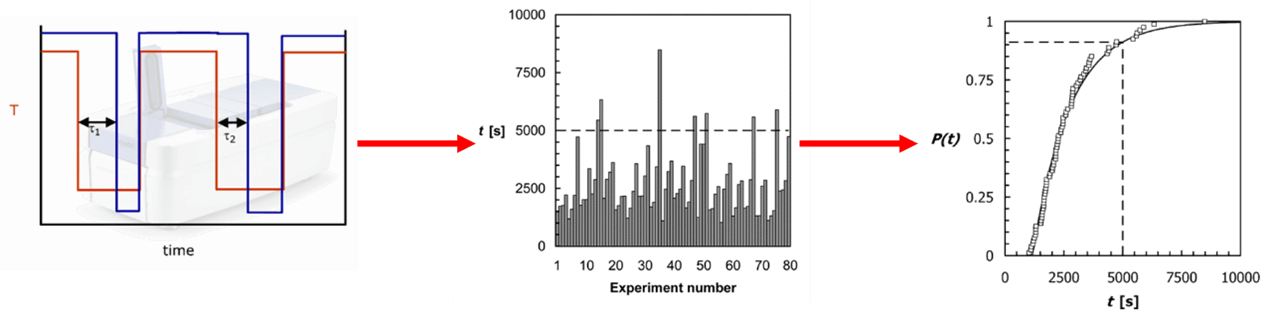
Figure 1: Processing of induction time data
Feedback control
Using the integrated Crystal16’s feedback control function, it is possible to dramatically reduce the time needed to collect the amount of data required to calculate the nucleation rate.
Induction time experiments can take several hours to collect a single data point. Complete data sets could take weeks. This can be reduced to only a couple of hours, through our automated feedback control system. The Crystal16 instrument with the feedback control functionality enables you to automatically detect when the compound is completely dissolved (clear point) or when it crystallizes (cloud point) and triggering the next temperature-controlled step. By making use of the feedback functionality offered by the Tehnobis Crystallization Systems software, experiment time can be reduced dramatically. Some case studies showed a time reduction from 70 hrs to 15 hrs.[iv]
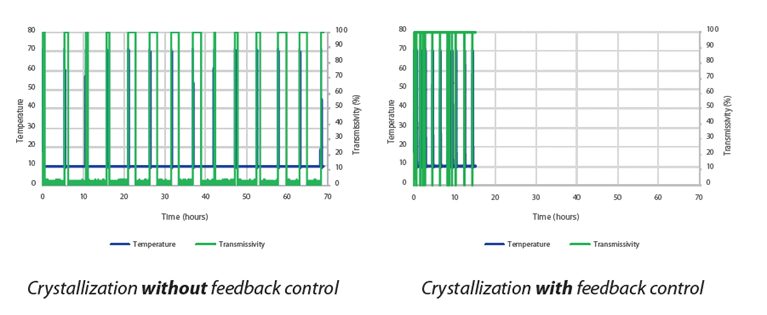
Figure 2: Induction time measurements with and without feedback control
Case study
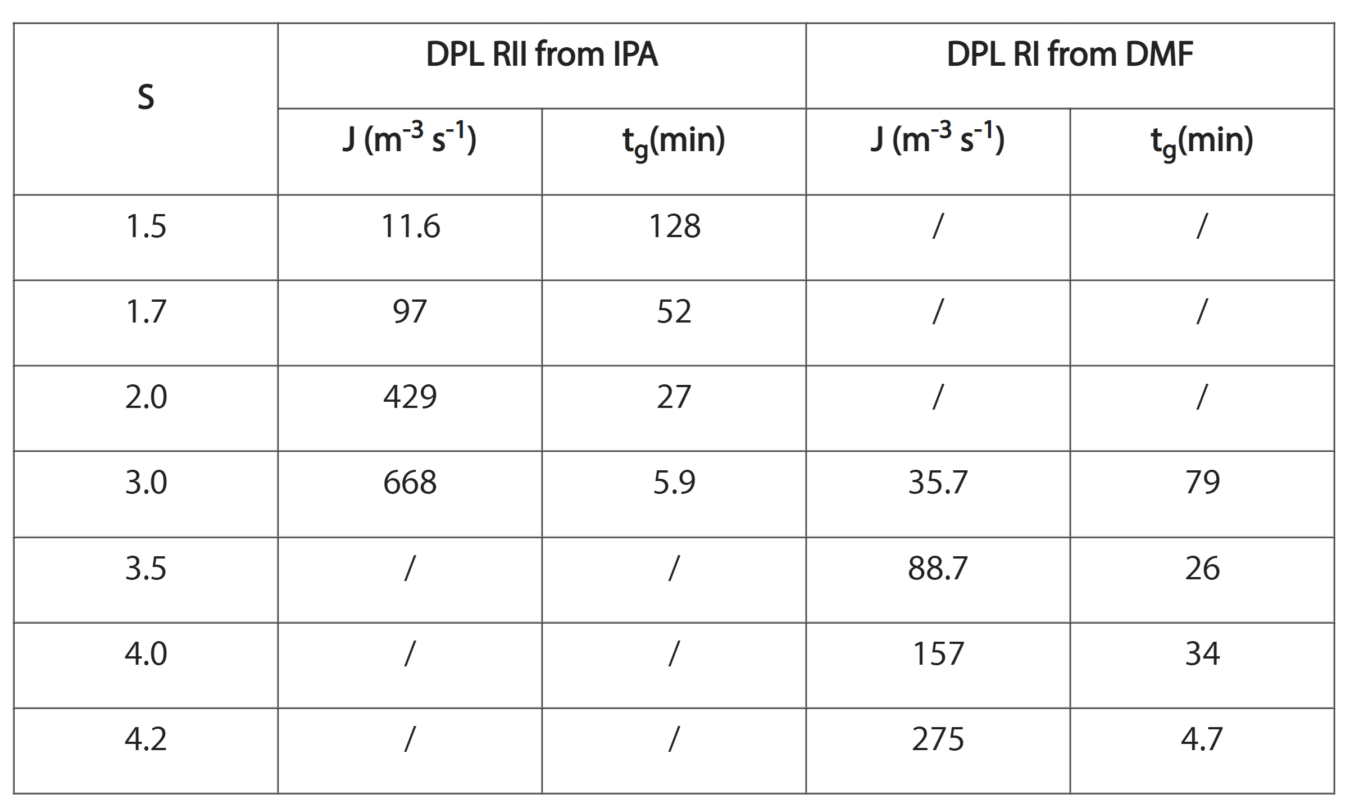
The nucleation rate of two polymorphs of Diprophylline (DPL) was investigated using the induction time method in IPA and DMF at different supersaturations. Samples were temperature cycled between 60 and 25 °C, holding at 25 °C until crystallization was detected.
Induction time was then measured between when the crystallization temperature was reached, and crystallization was detected. Using this method cumulative probability time plots were made and nucleation rates calculated form the fitting. These results showed the nucleation rate of Form RII to be much higher in IPA than Form RI in DMF. Additional analysis was done using the nucleation rate to calculate thermodynamic and kinetic nucleation factors.
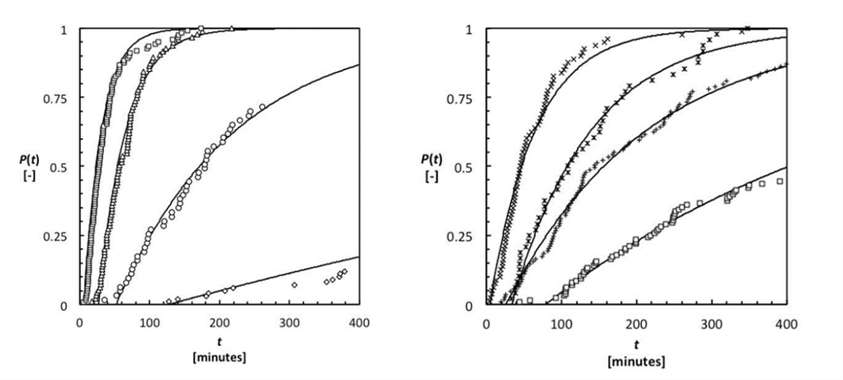
Figure 3: Cumulative probability plots of induction time measurment at different supersaturations of diprophylline IPA (Left), and DMF (right)
Using the Crystal16 it has never been easier to measure nucleation rates, with the first commercially available software that collect, plots and analyses induction times data. Additionally, the feedback control software can significantly reduce the time needed to run induction time experiments, resulting in nucleation rates in a couple of hours.
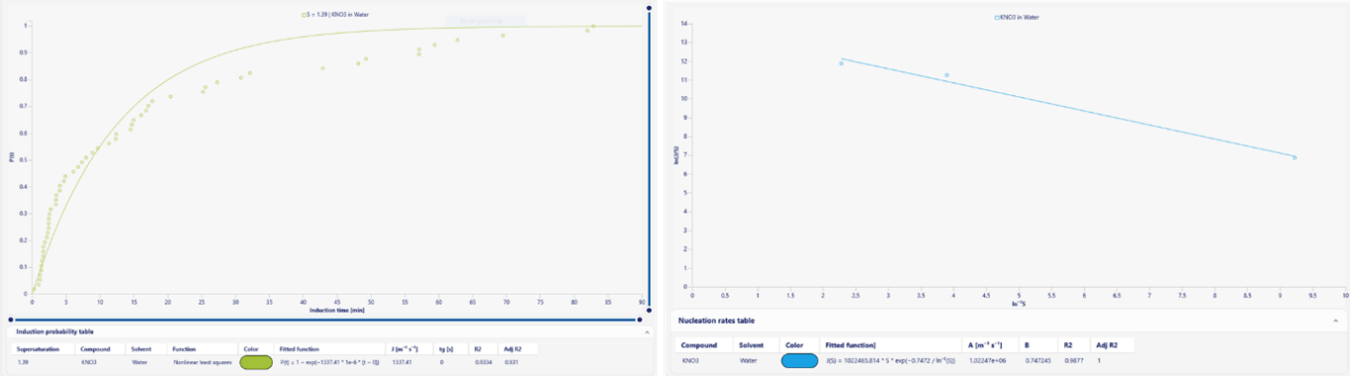
Figure 4: Nucleation rate fitting for Potassium Nitrate (KNO3) analyzed using the Crystal16 software
References
[i] Turnbull, David (1952). Kinetics of Solidification of Supercooled Liquid Mercury Droplets. The Journal of Chemical Physics, 20(3), 411–. doi:10.1063/1.1700435
[ii] Knezic, Dragutin; Zaccaro, Julien; Myerson, Allan S. (2004). Nucleation Induction Time in Levitated Droplets. The Journal of Physical Chemistry B, 108(30), 10672–10677. doi:10.1021/jp049586s
[iii] Jiang. S; and ter Horst. J, Crystal Nucleation Rates from Probability Distributions of Induction Times., Cryst. Growth Des. 2011, 11, 1, 256–261, https://doi.org/10.1021/cg101213q
[iv] R. R. E. Steendam, U. B. R. Khandavilli, L. Keshavarz and P. J. Frawley, Cryst. Growth & Des. 2019, 19, 8, 4483-4488
Curious to learn more?
Read more about this case study in the whitepaper "Calculate Nucleation Rate from Induction Time Measurements: Theory and Applications with Crystal16".