Since 2004, Crystallization Systems has helped scientists around the world with process analytical technology (PAT) based small scale equipment by providing solutions in crystallization development.
Both the Crystal16 and Crystalline instruments can be used as a strategy for the development of a crystallization process. PAT implemented at scales as small as milliliters in medium-throughput equipment, enables the early acquisition of key data to select the best crystallization approach.
The development of crystallization processes based on the Crystal16 and Crystalline as PAT is a strategy for:
- Identification of best solvent media through high throughput solubility screens;
- Identification of best approach to crystallization via small scale PAT;
- Understanding the properties of the solid/solvent system through PAT.

Normal initial experimental phases of crystallization process development are commonly carried out at very small scales, typically using 1-5mL vessels. The objective of these early phases of process development are to select a solvent based on solubility and solid form. These activities are commonly conducted in reactor systems, such as the Crystal16 and Crystalline.
However, for the development, validation and optimization of crystallization process models, this data is usually not utilized and the selected solution system is probed experimentally and more quantitatively at much larger scales, typically between 100 – 1000mL. A more quantitative usage of the data generated at small scale for the development of process models which may significantly reduce the number of larger scale experiments required, would aid in addressing the increasing constraints on time and materials in pharmaceutical development.
Solubility and metastable zone width (MSZW) experiments are routinely conducted with both experimental systems, with clear points (indicating the point of dissolution) and cloud points (indicating the on-set of nucleation in solution) utilized to indicate the MSZW for a given cooling rate, agitation rate and solute composition. Through turbidity and temperature measurements, both experimental systems provide the ability to quantitatively determine MSZW and therefore, the primary nucleation kinetics of the solution system.
In addition, the Crystalline instrument also has the added measurement capabilities of particle view imaging, providing a number based representation of the particle size distribution (PSD), and Raman modules for concentration and solid form monitoring. In addition,the in-situ image analysis capabilities can be enormously beneficial in terms of aiding process understanding, providing crucial information for crystallization mechanism and model discrimination activities. The through the vial probes also lead to no cross contamination and no interference in the crystallization environment.
All of these techniques are integrated into a small reactor with overhead stirring and refluxing capabilities, which can qualitatively mimic the likely vessel configurations at larger scales.
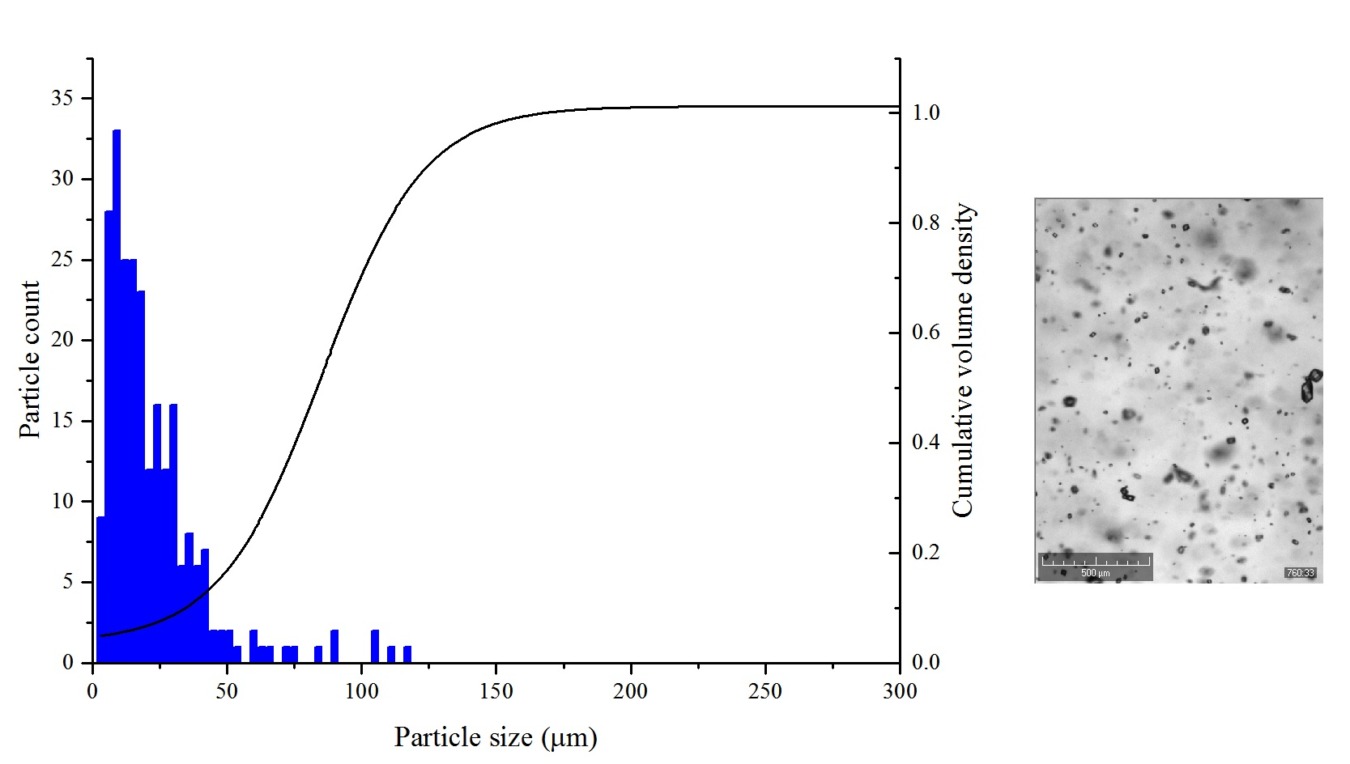
A study performed at CMAC in collaboration with PSE attempted to show that development, validation and optimization of crystallization process models which are done experimentally at much larger scales, may be succesfully performed at lower scale and significantly reduce the number of larger scale experiments required.
Data from paracetamol and 3-methyl-1-butanol solutions at the micro-scale was used to estimate the crystallization kinetics of the model, including crystal growth and primary nucleation, enabling model development, as well as mechanism discrimination. The final predictions of the developed process model were compared with a previously developed and validated process model, which employed larger scale 1L scale experiments.
A key outcome of this work is an adapted process development workflow for the design, model validation and optimization of processing models for crystallization systems. This workflow enables crystallization process development with an order of magnitude lower demands for materials. In particular raw API, which may not be available in the early stages of process development. In addition, the design space for the process can be assessed early on, such a process robustness and viability of continuous processing, with less dependence on larger scale, more material intensive experiments.
As a result, by utilizing commonly available micro-scale process data the material demands and requirements for larger scale experiments can be significantly reduced, leading to a step change in pharmaceutical process development efficiency and productivity.
References
- Cameron J. Brown, Niall Mitchell, Micro-scale process development and optimization for crystallization processes, Poster at ISIC Dublin 2017.
- Melissa Birch, Steven J. Fussell, Paul D. Higginson, Neil McDowall, and Ivan Marziano, Towards a PAT-Based Strategy for Crystallization Development, Organic Process Research & Development, Vol. 9, No. 3, 2005, 360-364.
Curious to learn more?
Learn about the Crystal16 and Crystalline products for fully controlled crystallization studies.