The successful development and commercialization of an active pharmaceutical ingredient requires adequate bioavailability and stability. Ideally, a newly developed compound should have BCS (Biopharmaceutics Classification System) class 1: the compound is highly soluble and highly permeable. However the reality is that most compounds in early development are not BCS class 1. If during the discovery phase, a compound is classified as BCS class 2, 3 or 4, there is a great need to improve the solubility and/or permeability. Among the five key physicochemical screens in early compound screening, pKa, solubility, permeability, stability and lipophilicity, poor solubility tops of the list of undesirable compound properties. As permeability is related to the actual active pharmaceutical ingredient itself and not its formulation form, salt screening is not an option for BCS class 3 compounds, but it should be used to improve the bioavailability for BCS class 2 and 4 compounds.
Conceptually, solubility is an easy parameter to measure but its meaning and concept of use is often different for discovery and development scientists and this can be a source of misunderstandings and controversy. In a broad sense, solubility is defined as the equilibrium amount of a crystalline compound that can be dissolved in a specific solvent system at the given process conditions, of which the temperature is often the most influential parameter. Measuring the solubility requires accurate control of temperature and composition in liquid and solid phase, preferably with the capability to generate a vast amount of data in a short period of time.
The Crystal16 and Crystalline provide the ideal tools to efficiently gather and analyze solubility data.

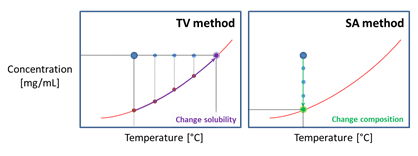
When solubility data is required at constant temperature, which is often the case in multicomponent mixtures. Solvent Addition (SA) is the method of choice. In the Crystalline, 8 measurements may be conducted simultaneously. Solvent is added to the vials over time using the syringe pumps. The cameras of the Crystalline clearly capture the thinning of the suspension upon the addition of solvent, until no crystals are detected anymore.

The Temperature Variation (TV) method is the most suitable method for determining the temperature dependent solubility line of a compound in a solvent. With the Crystal16, one can perform 16 solubility measurements at 1 mL scale. Upon heating a suspension of known composition, the temperature at which all crystals are dissolved marks a point on the solubility line. After a recrystallization step through cooling, the measurement can be repeated. An additional benefit of these cyclic measurements is that during the cooling stage the temperature at which the first crystals reappear can be recorded as the cloud point. The collection of cloud points give the metastable zone width (MSZW), which is used to determine the operation range of the process and indicates the tendency towards primary nucleation.
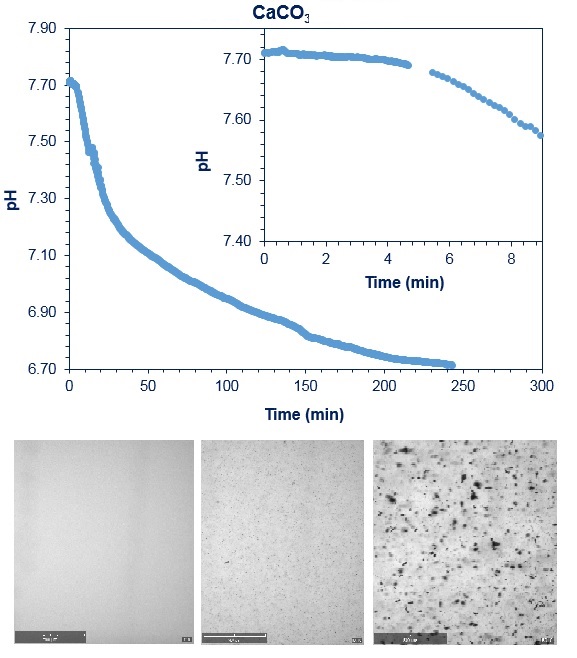
Buffered solubility also termed apparent solubility referring to solubility at a given pH, measured in a defined pH-buffered system is now possible on the Crystalline instrument by making use of the newly developed pH set-up. You are not only able to determine pH dependent solubility, but also visualize when crystallization occurs with change of pH. A recent study performed at IHE Delft Institute for Water Education by Dr. Sergio Salinas Rodriguez and his PhD student Muhammad Nasir Mangal showed that the pH dependent crystallization of calcium carbonate can be easily followed with the Crystalline Instrument. A drop of 0.02 in pH units was easily corelated with calcium carbonate crystallization and detected by the real time particle imaging available on the Crystalline instrument.
Curious to learn more?
Check out our whitepaper "Solubility: Importance, Measurements and Applications".