Challenges and possible solutions
The majority of active molecules in the pharmaceutical sector are oral formulations. However, a growing number of these molecules demonstrate poor aqueous solubility. Therefore, Strategies are needed to improve the solubility for the active molecule to be effective in the body.
Amorphous solid dispersions are increasingly seen as a viable way of achieving this. The Crystal16 is multi-screening platform that allows for screening of different amorphous solid dispersions as well as understand polymer properties such as upper critical solution temperature (UCST) and lower critical solution temperature (LCST).
Improving solubility: stabilizing the amorphous state
Some compounds just do not have the required solubility or dissolution rate and often they are killed in the development process due to their high energy state. Amorphous materials are seen as a solution to this problem, because of their high energy state which gives rise to improved dissolution rates and solubility over the crystalline forms.
However, because of this higher energy state, crystallization of the amorphous state is common during storage from elevated supersaturation relative to the crystalline state once dissolved compromising the improved solubility of the formulation. Amorphous solid dispersions (ASD) are designed to overcome this problem by stabilizing the amorphous state of the active ingredient in a polymer matrix.
The Crystal16 offers an invaluable tool for the automation of amorphous solid dispersion screening, enabling the fast and effective identification of new solid dispersions of compounds. With 16 reactors at a volume of 1mL, the instrument enables scientists to determine solubility curves and assess dispersion stability and critical solution temperatures.

Amorphous solid dispersions
ASD is a solid dispersion in which an amorphous active ingredient is dispersed within a matrix, typically a polymer. As there is no energy required to break the crystal lattice of the active ingredient, the higher solubility and faster dissolution are obtained compared to the crystalline form.
However, the properties of the polymers used can have a significant effect on the final properties of the formulation, such as hydrophilic polymers improving the wettability of hydrophobic molecules [1]. Therefore screening of the effectiveness of ASD needs to be undertaken to understand the properties of the formulation.
Amorphous solid dispersions screening may be performed with the Crystal16 in an automated, controlled and flexible manner. The screening may take into account, for instance, the effect of drug loading (e.g. 5, 10, 15 and 25%), type of solvent (binary or ternary systems may be also considered), and temperature profile on the formation of solid dispersions. This way, a wide set of small-scale experiments using a minimal amount of compound can be performed. For example, in one run with the 16 reactors of the Crystal16, one can check the effect of two solvents, two drug loads, and 4 polymers on the formation of solid dispersions for an active ingredient.
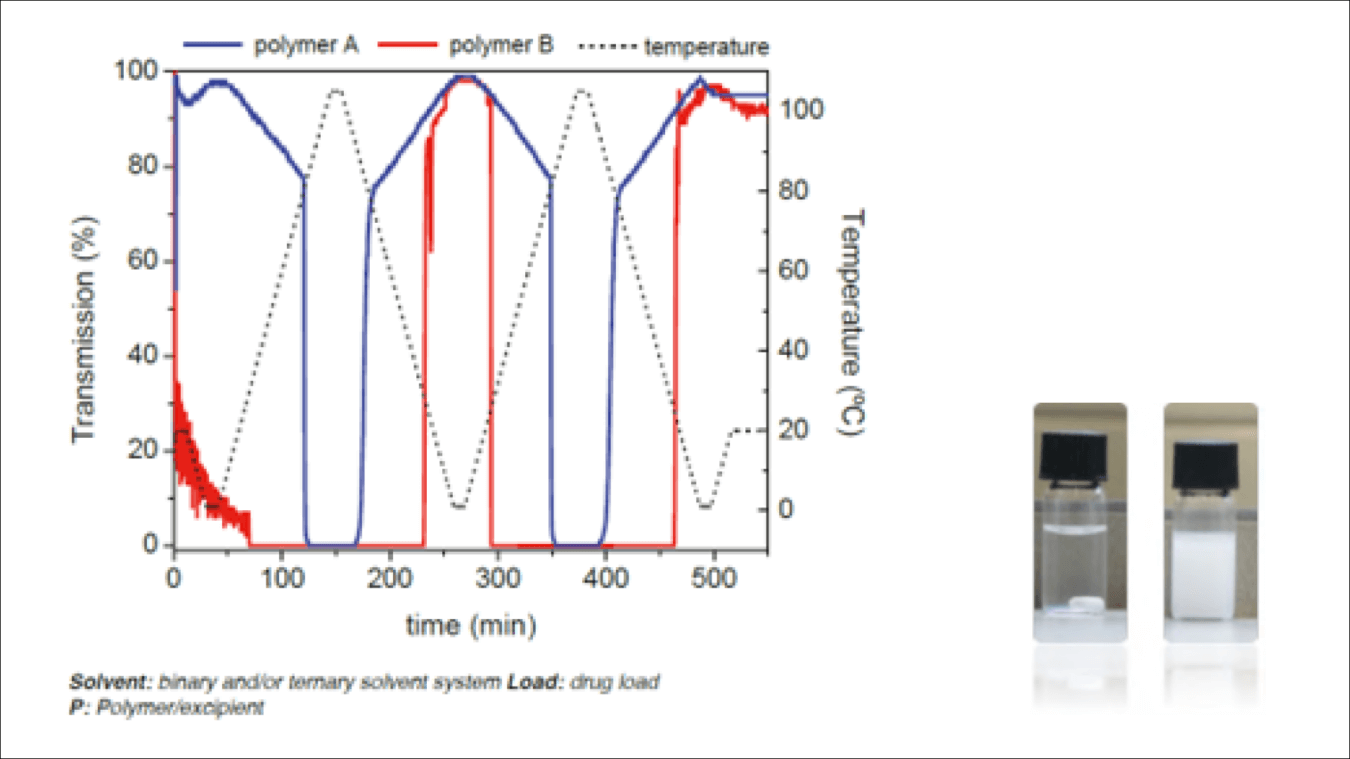
Investigating LCST and UCST
Polymer solutions sometimes show a complex thermal behavior, driven mostly by entropic forces. Some polymers show a typical thermal behavior, dissolving at high temperatures and crashing out upon cooling. In this case, the Tclear point denotes the upper critical solution temperature (UCST). In other cases, however, such as those for some PEG and polyoxazoline derivates, the molecules have an inverse behavior and crash out of the solution at higher temperatures. This is due to the favorable entropic effect of losing the solvation sphere of the polymer, and this temperature is called the lower critical solution temperature (LCST). This phenomenon can be used to develop controlled drug delivery systems and formulations close to or at body temperature.
References
[1] Vasconcelos T, Sarmento B, Costa P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov Today 2007;12:1068e75.
Curious to learn more?
Download the Application Note on "Improving Solubility: Stabilizing the Amorphous State".