Introduction
Anti-solvent is widely used in the pharmaceutical industry to improve the solid-state properties of pharmaceutical substances, such as modification of crystal formation, particle size distribution, and yield. Therefore, it becomes critical to understand the effect of anti-solvent experiment conditions on the Solution Mediated Polymorphic Transformation (SMPT) in pharmaceutical industries.
Experiments
Anti-solvent crystallization experiments were performed using Crystalline. Different volumes of anti-solvent added were 0.5 ml, 1 ml, and 2 ml. Additionally, to check the effect of flow rate, water was added to the solution at different flowrates of 10 ml/ hr and 30 ml/hr. Images at 1s intervals were recorded during the experimental run to examine the crystal morphology. After the completion of the experiment, the crystals formed were filtered, dried, and then weighed to calculate the actual yield.
Results & Discussion:
The addition of water as an anti-solvent reduces the solubility of CBZ in the solution, which generates high supersaturation conditions, resulting in the nucleation of metastable polymorphs (form II) because of lower interfacial energy. The nucleation kinetics chooses the formation of a specific form II with lower interfacial energy for the solute to crystallize. However, thermodynamics plays a critical role in leading to the nucleation of stable form III at lower supersaturations (Figure 1).

Figure 1: Nucleation of CBZ during anti-solvent crystallization at different anti-solvent
During the anti-solvent crystallization experiments, there was a substantial increase in the yield as compared to the theoretical yield from cooling crystallization, summarized in Table 1. With the increase in the antisolvent: solvent ratio, the yield significantly increased from 47.40% to 82.04%.
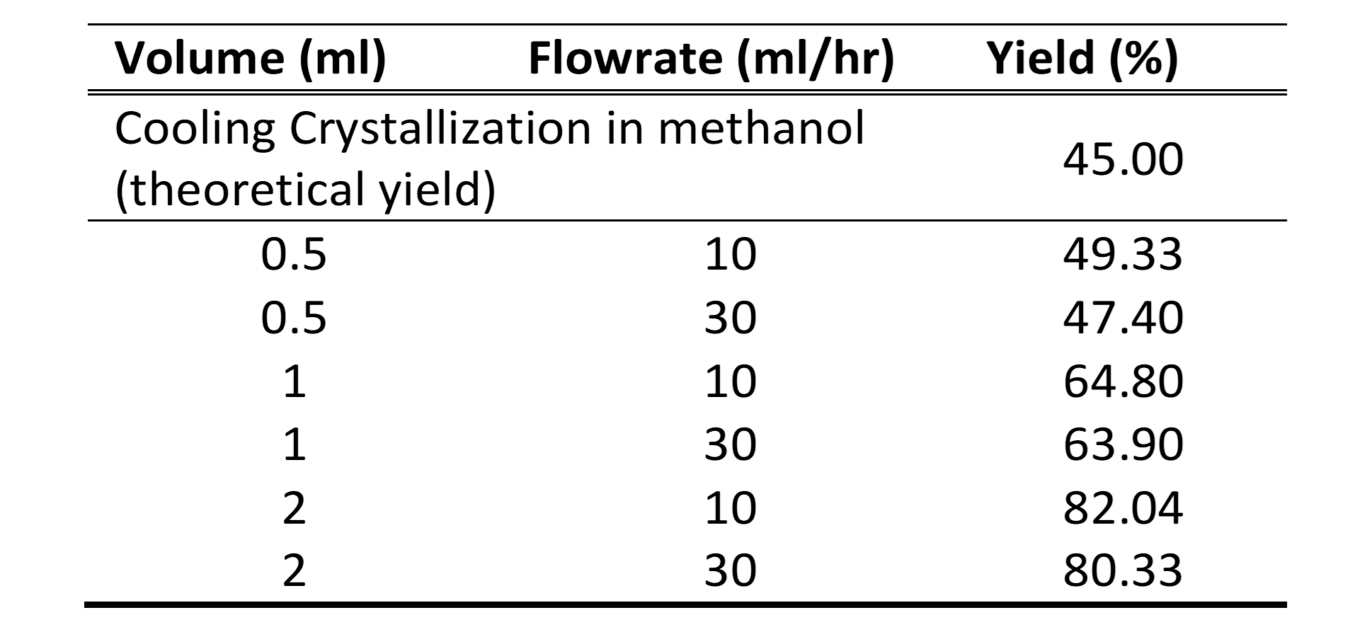
Table 1: Yield (%) of carbamazepine from anti-solvent crystallization
The morphologic transformation of CBZ crystals from nucleating as metastable form II into stable polymorphic form III with the addition of 0.5 ml water at 30 ml/hr during antisolvent crystallization was observed. Generation of sudden supersaturation with the addition of antisolvent nucleates metastable form II, seen as needle shape crystals, but with time they dissolve and transform into thermodynamically stable form III, seen as prismatic shape crystals in the Figure 2.
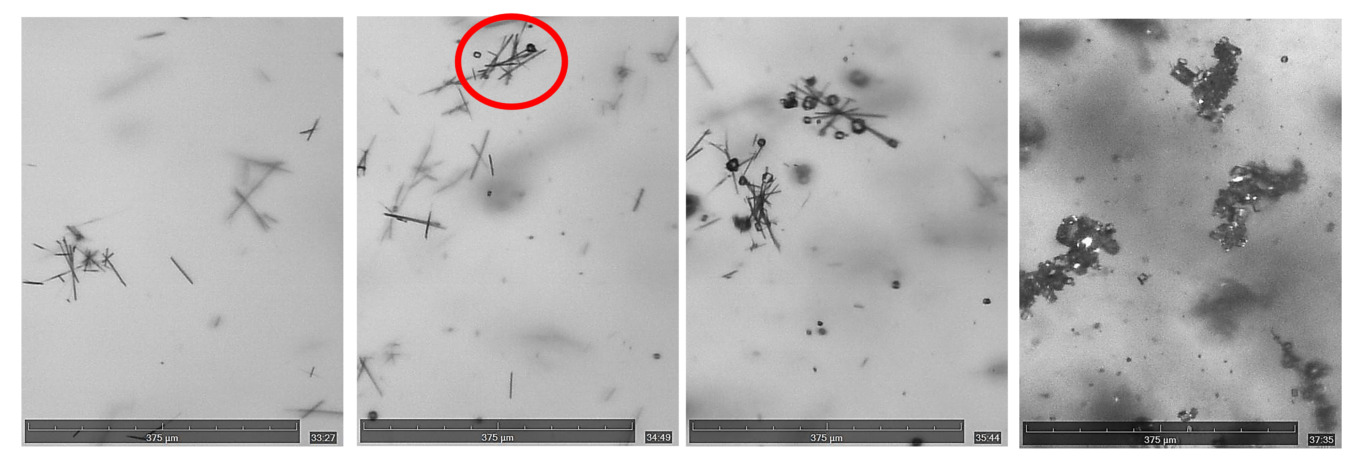
Figure 2: Images showing the transformation of metastable form II to stable form III during anti-solvent crystallization with addition of 0.5 ml water at 30 ml/hr flow rate
Conclusion
Crystalline facilitates the anti-solvent crystallization experiments with the usage of solvent dosage unit and accurately captures the in-situ polymorph transformation happening during crystallization processes.
Download Application Note
Curious to learn more?
Read the full scale experiment in the application note "Anti-solvent crystallization: witness the polymoprhic transformation with Crystalline"