Content overview
More than 80% of formulations are in the solid state, with most as tablets due to ease of transport, application and high patient compliance. Therefore, a well-designed and controlled crystallization process is paramount. However, this can be a time and material consuming process, as it can take multiple experiments to deliver the required results.
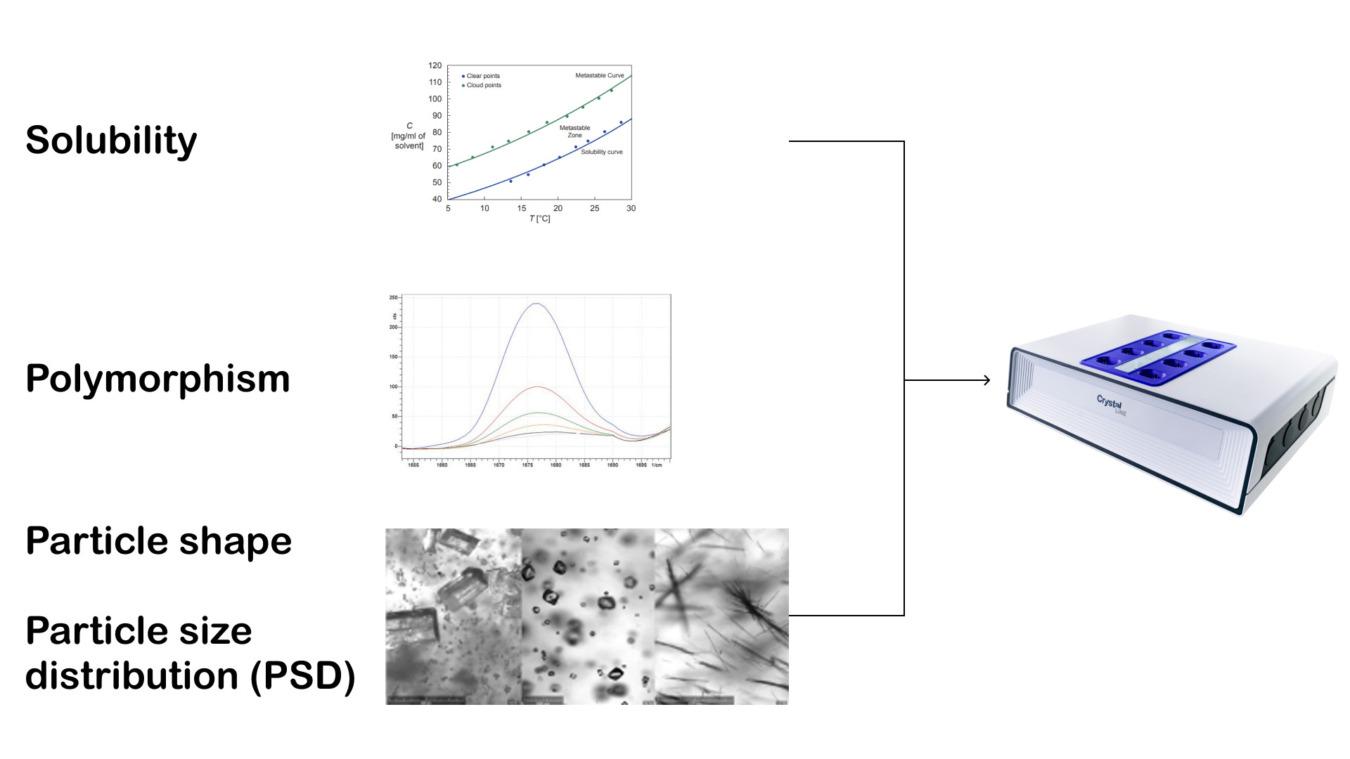
There are several particle parameters that are important to control during crystallization. Arguably the most significant are form purity, particle shape and particle size distribution - due to their legal and manufacturing implications.
This presentation explores how Crystalline V2 can be an ideal platform for crystallization development. We start with looking at solubility information, which is the foundation of any crystallization process. Then we demonstrate how, by using integrated Raman, you can track solid form. Finally, we discuss how it is possible to visualize crystal size and shape in real time with the use of AI particle detection, at the milliliter scale.
This presentation was presented during the webinar Enabling small-scale analysis during crystallization screening through a single platform. Download the webinar presentation slides using the form on this page or watch the webinar video recording in the Webinars section.