Content overview
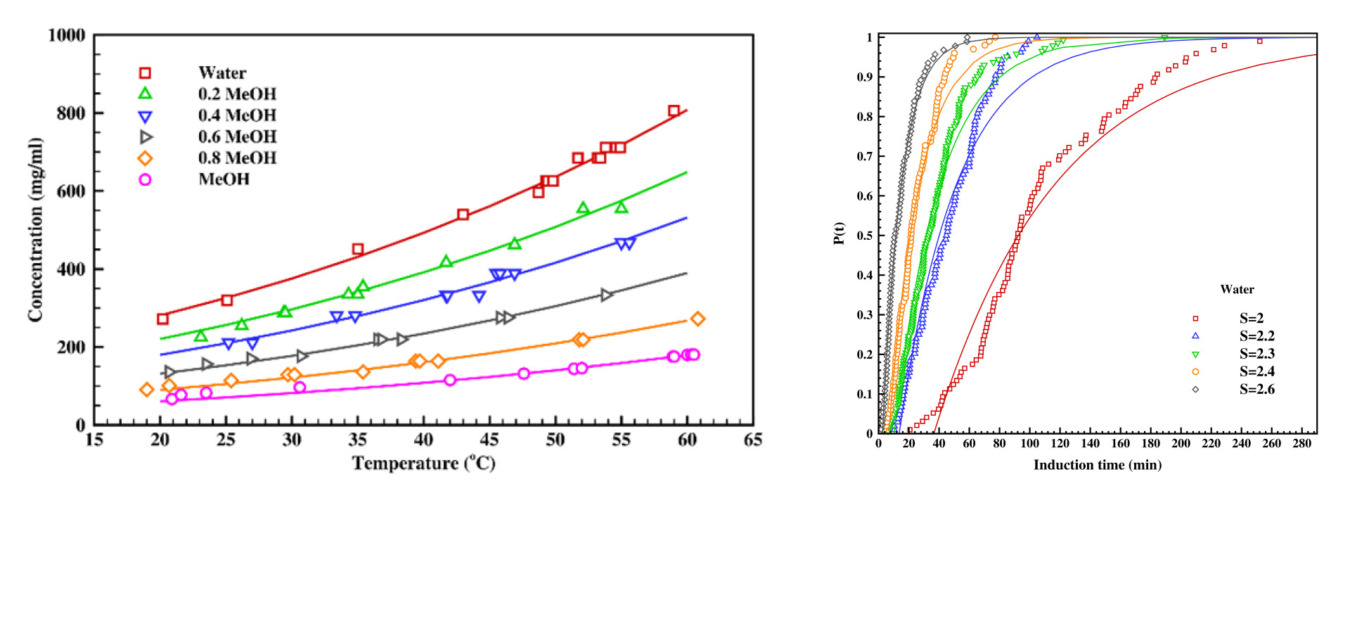
Crystallization is a necessary separation and purification step to produce any pharmaceutical products, and the choice of solvent plays a critical role in solution crystallization, affecting thermodynamic properties, kinetic parameters, and final crystal attributes. This experimental work focuses on the effect of various binary solvent systems of water-alcohols (methanol/ethanol/isopropanol) on the crystallization of Ascorbic acid (Vitamin C) in terms of solubility, nucleation rate, and crystal habit. Adding alcohols in binary solvent systems reduces the solubility, increases critical Gibbs free energy for nucleation, and transforms the crystal shape into elongated rod-shaped crystals.
This presentation was presented during the webinar Unlocking the Power of Solvent Selection: Influence on Solubility, Nucleation and Crystal Habit of Ascorbic Acid. Download the webinar presentation slides using the form on this page or watch the webinar video recording in the Webinars section.