
Prof. Dr. Joop ter Horst
Strathclyde University Glasgow
Measuring the Primary and Secondary Nucleation in Crystallization
Reliable information on solubility and crystallization kinetics is of utmost importance for a robust crystallization process design [1]. Commercial equipment is available to accurately measure the solubility of a solid [2]. Equipment such as the Crystal16 uses clear point temperature measurements in which the temperature at which the suspension turns into a clear solution upon slowly heating the sample. Knowing the solubility is key information in designing crystallization processes and helps for instance in selecting the crystallization method [3].
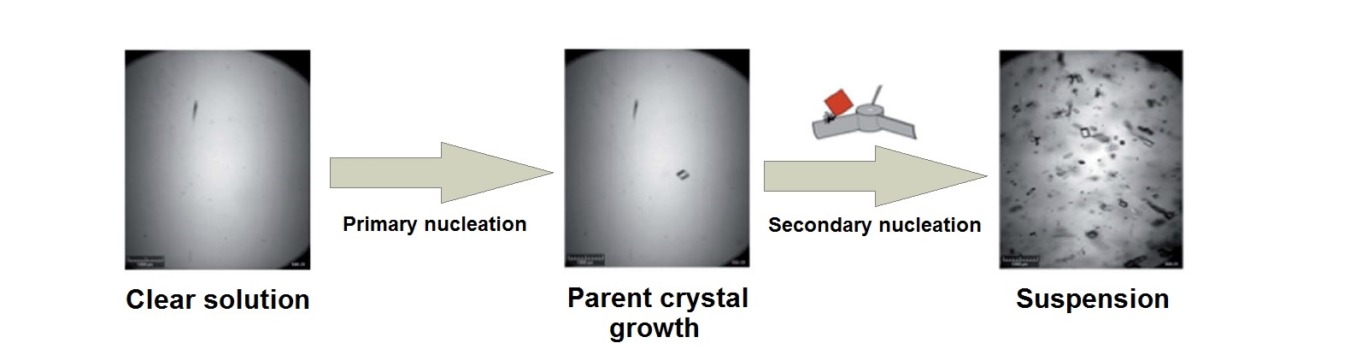
While the solubility defines the yield of the process, crystallization kinetics determine the process time. Primary crystal nucleation takes place from clear supersaturated solutions [4]. The induction time, the time it takes at constant supersaturation to detect crystals, captures information on this primary nucleation rate. Due to the stochastic nature of the nucleation process at specific supersaturation conditions a distribution of induction times can experimentally be determined, which, through a statistical analysis, results in a nucleation rate [5,6].
With aid of commercial equipment such as the Crystal16 such experimental induction time distributions can nowadays be routinely measured. The results show that nucleation rates for organic compounds from solutions are quite low. This may be due to either a slow attachment rate of solute molecules to the nucleus or a low concentration of heterogeneous particles.
Secondary nucleation occurs as a result of the presence of other large crystals of the same compound in a supersaturated suspension. Thus, in case of secondary nucleation parent crystals serve as catalyst for the formation of small new crystals. Using a single crystal seed technique the secondary nucleation rate can be measured in commercially available equipment such as the Crystalline. In this method the initial increase in particle count upon single crystal seeding a supersaturated solution is monitored. This allows establishing a secondary nucleation supersaturation threshold that can be used in the design of crystallization processes to either prevent or enhance secondary nucleation rates.
The availability of commercial equipment allows standardizing experimental approaches to obtain reliable data for a consistent and robust crystallization process design.
You can download the webinar presentation slides from our Publications section.

Strathclyde University Glasgow