
Rene Steendam
Project Leader Solid State Research (SSR), Ardena
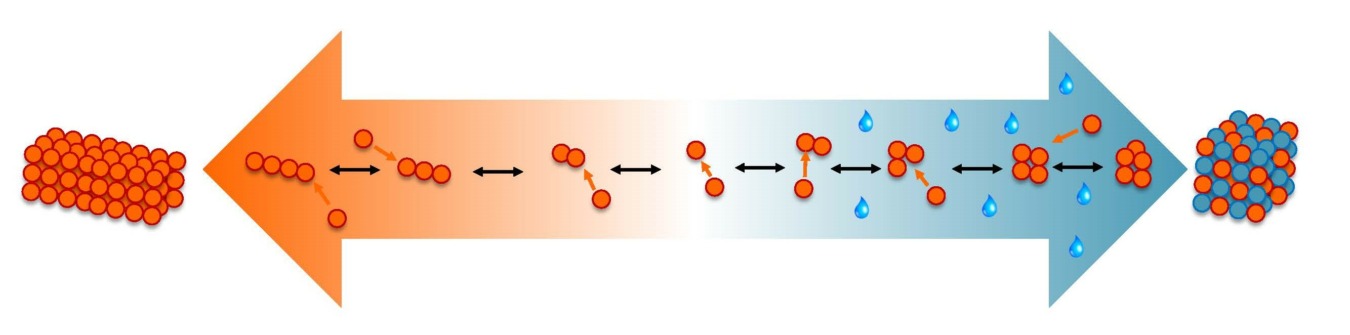
Crystallization from solution is widely used to purify and isolate active pharmaceutical ingredients (API). The formation pathway of crystals strongly depends on interactions between solvent molecules and molecules of the API. As a result, solution chemistry influences the final properties of the crystalline API such as particle shape, size and polymorphism.
Using analytical techniques, the ordering of molecules in solution can be experimentally measured and linked to the crystalline form. Furthermore, research into the early stages of crystal nucleation can be predicted using nucleation kinetics obtained from induction time measurements. Together, these approaches have provided insight into the roles of desolvation and molecular self-assembly in the crystallization of organic molecules.
In this webinar, Dr René Steendam presented a few examples in which solution-phase measurements as well as nucleation kinetics can be used to elucidate how molecules in solution become large ordered crystalline structures. The role of solvent choice in the crystallization of the blockbuster drug Pregabalin were presented in detail.
References
Watch the webinar video recording using the form on this page or download the webinar presentation slides from the Publications section.

Project Leader Solid State Research (SSR), Ardena