About the webinar
This webinar was brought to you in collaboration with APC.
Topic:
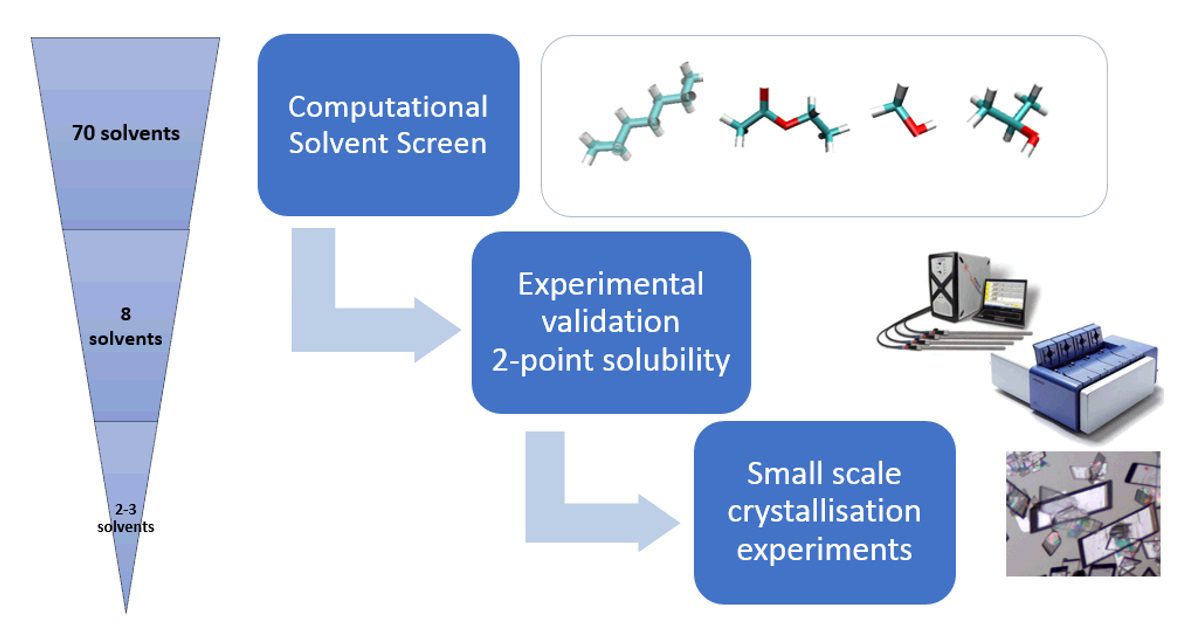
Choosing an appropriate solvent is the cornerstone of good crystallization development. The solvent system from which an active pharmaceutical ingredient (API) is crystallized, influences solubility, growth and nucleation kinetics, impurity purging, polymorph control, solvation propensity and crystal morphology. Despite the importance of this choice, in early phase development, solvents are typically chosen based on chemical intuition with only the impact of product yield considered. This can lead to insufficient purity, unfavourable particle size and serious processability issues in later phases of both drug substance and drug product development. At APC, using predictive modelling and smart experimentation, it is developed a platform that enables optimal solvent selection across eight criteria: yield, productivity, antisolvent efficiency, solvation and oiling propensity, impurity rejection, seedability and safety and environmental choices. APC's early stage crystallization workflow involves an initial in-silico screen that combines thermodynamic and quantum chemistry modelling followed by narrow experimental validation. Benefits of employing in-silico methods are multiple, such as expanding the number of neat solvents and mixtures computationally screened, while minimising material requirements, and avoiding incorrect thermodynamic choices for yield, form, solvation etc.
In this webinar, Dr Marko Ukrainczyk demonstrated APC's early phase crystallization process development strategy, through case studies of propriety API molecules, chosen as examples of their cooling and antisolvent crystallization workflows. The use of Raman spectroscopy was also demonstrated in the context of early form/solvates and impurity identification.
Watch the webinar video recording using the form on this page or download the webinar presentation slides from the Publications section.